


GLOBAL PRETERM BIRTH RATES – Hungary
Estimated # of preterm births: 8.84 per 100 live births (USA 9.56-Global Average: 10.6)
Source- WHO 2014- https://ptb.srhr.org/
Hungary is a landlocked country in Central Europe. Spanning 93,030 square kilometres (35,920 sq mi) of the Carpathian Basin, it is bordered by Slovakia to the north, Ukraine to the northeast, Romania to the east and southeast, Serbia to the south, Croatia and Slovenia to the southwest, and Austria to the west. Hungary has a population of 9.7 million, mostly ethnic Hungarians and a significant Romani minority. Hungarian, the official language, is the world’s most widely spoken Uralic language and among the few non-Indo-European languages widely spoken in Europe. Budapest is the country’s capital and largest city; other major urban areas include Debrecen, Szeged, Miskolc, Pécs, and Győr.
Hungary is a middle power in international affairs, owing mostly to its cultural and economic influence. It is a high-income economy with a very high human development index, where citizens enjoy universal health care and tuition-free secondary education. Hungary has a long history of significant contributions to arts, music, literature, sports, science and technology. It is a popular tourist destination in Europe, drawing 24.5 million international tourists in 2019. It is a member of numerous international organisations, including the Council of Europe, NATO, United Nations, World Health Organization, World Trade Organization, World Bank, International Investment Bank, Asian Infrastructure Investment Bank, and the Visegrád Group.
Hungary maintains a universal health care system largely financed by government national health insurance. According to the OECD, 100% of the population is covered by universal health insurance, which is free for children, students, pensioners, people with low income, handicapped people, and church employees. Hungary spends 7.2% of GDP on healthcare, spending $2,045 per capita, of which $1,365 is provided by the government.
Hungary is one of the main destinations of medical tourism in Europe, particularly for dentistry, in which its share is 42% in Europe and 21% worldwide. Plastic surgery is also a key sector, with 30% of the clients coming from abroad. Hungary is well known for its spa culture and is home to numerous medicinal spas, which attract “spa tourism”.
Source: https://en.wikipedia.org/wiki/Hungary

COMMUNITY

Exposure to Air Pollution and Emergency Department Visits During the First Year of Life Among Preterm and Full-term Infants
Original Investigation Environmental Health February 22, 2023 Anaïs Teyton, MPH1,2,3; Rebecca J. Baer, MPH4,5; Tarik Benmarhnia, PhD3; et alGretchen Bandoli, PhD1,5 JAMA Netw Open.2023;6(2):e230262. doi:10.1001/jamanetworkopen.2023.0262
Key Points
Question What is the association between fine particulate matter (PM2.5) exposure and emergency department (ED) visits during the first year of life, and are preterm infants more susceptible to PM2.5 exposure than full-term infants?
Findings In this cohort study of 1 983 700 infants, a positive association was observed between PM2.5 exposure and all-cause, infection-related, and respiratory-related visits. Preterm and full-term infants were most susceptible to having an all-cause ED visit during their fourth and fifth months of life.
Meaning These findings suggest that increased PM2.5 exposure was associated with an increased ED visit risk; thus, strategies aimed at reducing PM2.5 exposure for infants may be warranted.
Abstract
Importance Previous studies have focused on exposure to fine particulate matter 2.5 μm or less in diameter (PM2.5) and on birth outcome risks; however, few studies have evaluated the health consequences of PM2.5 exposure on infants during their first year of life and whether prematurity could exacerbate such risks.
Objective To assess the association of PM2.5 exposure with emergency department (ED) visits during the first year of life and determine whether preterm birth status modifies the association.
Design, Setting, and Participants This individual-level cohort study used data from the Study of Outcomes in Mothers and Infants cohort, which includes all live-born, singleton deliveries in California. Data from infants’ health records through their first birthday were included. Participants included 2 175 180 infants born between 2014 and 2018, and complete data were included for an analytic sample of 1 983 700 (91.2%). Analysis was conducted from October 2021 to September 2022.
Exposures Weekly PM2.5 exposure at the residential ZIP code at birth was estimated from an ensemble model combining multiple machine learning algorithms and several potentially associated variables.
Main Outcomes and Measures Main outcomes included the first all-cause ED visit and the first infection- and respiratory-related visits separately. Hypotheses were generated after data collection and prior to analysis. Pooled logistic regression models with a discrete time approach assessed PM2.5 exposure and time to ED visits during each week of the first year of life and across the entire year. Preterm birth status, sex, and payment type for delivery were assessed as effect modifiers.
Results Of the 1 983 700 infants, 979 038 (49.4%) were female, 966 349 (48.7%) were Hispanic, and 142 081 (7.2%) were preterm. Across the first year of life, the odds of an ED visit for any cause were greater among both preterm (AOR, 1.056; 95% CI, 1.048-1.064) and full-term (AOR, 1.051; 95% CI, 1.049-1.053) infants for each 5-μg/m3 increase in exposure to PM2.5. Elevated odds were also observed for infection-related ED visit (preterm: AOR, 1.035; 95% CI, 1.001-1.069; full-term: AOR, 1.053; 95% CI, 1.044-1.062) and first respiratory-related ED visit (preterm: AOR, 1.080; 95% CI, 1.067-1.093; full-term: AOR,1.065; 95% CI, 1.061-1.069). For both preterm and full-term infants, ages 18 to 23 weeks were associated with the greatest odds of all-cause ED visits (AORs ranged from 1.034; 95% CI, 0.976-1.094 to 1.077; 95% CI, 1.022-1.135).
Conclusions and Relevance Increasing PM2.5 exposure was associated with an increased ED visit risk for both preterm and full-term infants during the first year of life, which may have implications for interventions aimed at minimizing air pollution.
Source:https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2801735

Considerations for Reducing Maternal Mortality
Elizabeth Filipovich, MPH
Maternal mortality in the United States is on the rise and has been for the past several decades. This trend stands out as other high-income countries, like the United Kingdom and Canada, have lower maternal mortality rates. Birthing people in the United States now experience worse mortality rates than the prior two generations. Maternal mortality ratios, or deaths per 100,00 live births, are used to illustrate the massive racial disparities among birthing people. Non-Hispanic Black birthing people have pregnancy-related mortality rates nearly 3x that of their white counterparts.
The Centers for Disease Control defines maternal mortality as “the death of a woman during pregnancy, at delivery, or soon after delivery.” Maternal deaths are further divided into two categories: pregnancy-related and pregnancy-associated deaths. Pregnancy-related deaths are defined as “the death of a woman while pregnant or within one year of the end of pregnancy, regardless of the outcome, duration, or site of pregnancy, from any cause related to or aggravated by the pregnancy or its management but not from accidental or incidental causes.”
Pregnancy-associated but not related deaths are “the death of a woman while pregnant or within one year of pregnancy from a cause or cause unrelated to pregnancy. Often, when maternal mortality is researched and discussed, the body of work emphasizes pregnancy-related deaths. For example, the statistics used in the above paragraph reference pregnancy-related deaths exclusively. However, a better understanding of factors contributing to many accidental, pregnancy-associated but not related deaths is essential for effective methods to reduce the number of maternal deaths in the United States, regardless of cause or manner of death.
Well-documented maternal death causes include hemorrhage, cardiomyopathy, or other cardiac causes, and worsening underlying conditions or other medical causes often deemed pregnancy-related. Equally important are other causes of death, including accidental poisonings or overdoses, maternal suicides, or homicides. These are pregnancy-associated, not related, or not directly caused or exacerbated by pregnancy. The many touchpoints of care in the perinatal period provide opportunities for intervention and opportunities for improved perinatal care, particularly for birthing people who have a history of substance use disorder (SUD), history of anxiety, depression, or other mood disorders, or families who may be at risk for violence, instability, or other significant hardship.
Statewide and local Maternal Mortality Review Committees (MMRC) are convened to examine maternal death trends by comprehensively reviewing deaths that occur during or within one year of pregnancy. MMRCs are multidisciplinary and include representatives from a spectrum of perinatal care providers, including public health, obstetrics, maternal-fetal medicine, pediatrics, nursing, midwifery, community health organizations, mental and behavioral health, and patient/family advocacy groups. MMRCs meet to discuss cases and collaboratively create evidence-based recommendations to prevent future deaths. MMRCs provide critical evidence for legislatures, health systems, and public health leaders to endorse safety bundles and new laws to prevent future deaths.
While MMRCs retrospectively review maternal deaths to understand preventable causes of these deaths further, providers and clinicians across all disciplines, as well as the public, can proactively impact the alarming rate of maternal deaths in this country. Neonatal care providers have a critical role. Despite becoming increasingly standard practice to have postpartum follow-up visits before four weeks postpartum, this is not universally implemented. Even if a postpartum follow-up is scheduled, not all birthing people attend a follow-up visit, as evidenced by several studies documenting that 11-46% do not attend a postpartum visit. However, well-child visits are very well attended by postpartum people. By capitalizing on the touchpoint of the well-child visits, providers capture an opportunity for assessment and potential referral or intervention.
Neonatal providers can contribute to reducing maternal mortality in several ways. Pediatric and family providers are often left out of the conversation, but the reality is that many providers for infants have more touchpoints with birthing people in the postpartum period than their prenatal providers. Pediatric visits for neonates and infants provide the opportunity for intervention that begins with a thorough assessment of the birthing person and include awareness of resources available to provide to patients, as well as understanding that wellness is facilitated by a host of factors extend beyond the physical health of the patient.
The scope of this newsletter article is not broad enough for the depth of discussion, but rather draws attention to how social determinants of health contribute to maternal deaths and how providers can continue to care for their patients by addressing them. Providers should attempt to understand the environment of each family. By exploring significant relationships, one can understand the birthing person’s support systems, the likelihood of experiencing violence, housing circumstances, income stability, etc. By connecting identified birthing persons to support services and resources and following up on successive pediatric visits, perinatal providers can reduce maternal mortality. For more information on perinatal mood disorders, perinatal substance use, and many other resources for providers and families, please visit NationalPerinatal.org.
Source:http://neonatologytoday.net/newsletters/nt-jan23.pdf

Want to grab a little sunshine! Take a listen to this fun song!
VALMAR ft. Szikora Robi – Úristen

Valmar is a popular Hungarian artist/band. Szikora Róbert – Hungarian singer and songwriter.

Optimizing Temperature of Preterm Infants in the Delivery Room
Preventing heat loss in infants less than 1500 grams and/or less than 30 weeks’ gestational age.



Bundle care approach
Preterm Baby Package Jan 22, 2023

HEALTHCARE PARTNERS

Recognizing Our Biases, Understanding the Evidence, and Responding Equitably
Application of the Socioecological Model to Reduce Racial Disparities in the NICU-McCarty, Dana B. DPT, PT Editor(s): Christine A., Fortney PhD, RN, Section Editor-Advances in Neonatal Care 23(1):p 31-39, February 2023.
Abstract
Background:
Implicit bias permeates beliefs and actions both personally and professionally and results in negative health outcomes for people of color—even in the neonatal intensive care unit (NICU). NICU clinicians may naïvely and incorrectly assume that NICU families receive unbiased care. Existing evidence establishing associations between sex, race, and neonatal outcomes may perpetuate the tendency to deny racial bias in NICU practice.
Evidence Acquisition:
Using the socioecological model as a framework, this article outlines evidence for racial health disparities in the NICU on multiple levels—societal, community, institutional, interpersonal, and individual. Using current evidence and recommendations from the National Association of Neonatal Nurses Position Statement on “Racial Bias in the NICU,” appropriate interventions and equitable responses of the NICU clinician are explored.
Results:
Based on current evidence, clinicians should reject the notion that the social construct of race is the root cause for certain neonatal morbidities. Instead, clinicians should focus on the confluence of medical and social factors contributing to each individual infant’s progress. This critical distinction is not only important for clinicians employing life-saving interventions, but also for those who provide routine care, developmental care, and family education—as these biases can and do shape clinical interactions.
TABLE 1. – NANN’s Racial Disparity in the NICU Position Statement Recommendations
| Elevate awareness of racial disparities, inclusion, and cultural sensitivity by providing education in cultural competence, presenting published research on the issues, and having open discussions about the topics. |
| Encourage diversity in the workforce. |
| Examine personal bias and beliefs, some of which may be unconscious. Be self-aware and open to feedback and observations from others. |
| Examine individual NICU statistics to evaluate significant trends in gestational age, race, and patient outcomes. |
| Invite families to participate in the culture of the NICU by involving a diverse team of parents on committees, such as a quality improvement committee. |
| Regularly use interpreters when caring for families who do not speak English. Relying on other family members to interpret for parents may contribute to misinformation and a lack of appropriate education. |
| Provide written and electronic information in multiple languages whenever possible. |
| Consider all discharge requirements and available resources to transition families to the home environment. |
| Advocate for racial awareness and equality in your hospital and community. Connect with hospital administrators, community leaders, and elected officials to discuss health outcomes of racial disparities, and advocate for resources that positively impact the social determinants of health affecting maternal and infant health. |
FULL ARTICLE:
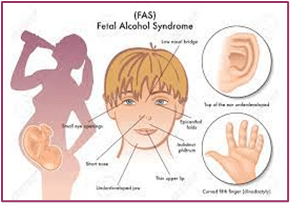
A hidden epidemic of fetal alcohol syndrome
New legislation could help bring awareness and resources to prevention, diagnosis, and treatment of fetal alcohol spectrum disorders-By Kirsten Weir Date created: July 1, 2022

Stress and alcohol use often go hand in hand, a concerning pattern on the heels of the COVID-19 pandemic. Researchers have found that alcohol use increased sharply during the pandemic, and there is some evidence that those patterns were present among pregnant women as well, said Ira Chasnoff, MD, a pediatrician and fetal alcohol spectrum disorder (FASD) researcher at the University of Illinois College of Medicine in Chicago. Experts worry that the trend could result in more babies being born with damage from prenatal alcohol exposure.
Even before the pandemic, FASD was a significant problem. Experts estimate that 2% to 5% of U.S. schoolchildren—as many as 1 in 20—may be affected by prenatal alcohol exposure, which can cause complications with growth, behavior, and learning. The effects on individuals and families, as well as the economic costs, are substantial.
Yet support for FASD research and services is limited. The National Institute on Alcohol Abuse and Alcoholism funds innovative research on FASD, said Christie Petrenko, PhD, a clinical psychologist and research associate professor at Mt. Hope Family Center, University of Rochester, and codirector of the FASD Diagnostic and Evaluation Clinic there. But a Substance Abuse and Mental Health Services Administration (SAMHSA)–funded FASD Center for Excellence program was shuttered in 2016, leaving a big gap between the research being done and practical solutions for children and families affected by FASD, she said. Now, there’s a bipartisan bill before Congress, the FASD Respect Act, which would support FASD research, surveillance, and activities related to diagnosis, prevention, and treatment. (APA has endorsed this bill.)
Such attention is sorely needed, and psychologists have a significant role to play in diagnosis, prevention, and treatment, Petrenko said. “Families are desperate for support.” Yet many people with FASD haven’t even received an accurate diagnosis, let alone appropriate treatments.
Clinicians should be aware that FASD often overlaps with mental health symptoms. These problems begin in early childhood and exist through adulthood, as described by Mary O’Connor, PhD, ABPP, founder of the UCLA Fetal Alcohol Spectrum Disorders Clinic (Current Developmental Disorders Reports, Vol. 1, No. 1, 2014). Her research has also found a higher incidence of suicidal ideation and behavior in adolescents with FASD (Birth Defects Research, Vol. 111, No. 12, 2019). And many adults with FASD who have mental health disorders aren’t getting treatment, said Susan Stoner, PhD, a research associate professor at the University of Washington School of Medicine and director of the Washington State Parent-Child Assistance Program, a program for pregnant and parenting women with substance use disorders (Alcoholism: Clinical and Experimental Research, Vol. 46, No. 2, 2022). “We found those with less severe FASD tend to have worse mental health than those with more severe FASD, which might be because those with more severe FASD are more likely to have a diagnosis and more likely to get support,” she said.
Understanding FASD
Many variables determine whether an infant will be born with FASD and how severe the disorder will be. Such factors include how much a pregnant person drinks, the rate at which they metabolize alcohol, and the stage of fetal development during alcohol exposure. “There are too many variables at play to estimate a safe level of drinking during pregnancy,” Stoner said. “The safest amount of alcohol during pregnancy is zero.”
Prenatal alcohol exposure can result in several conditions that fall under the FASD umbrella. These include fetal alcohol syndrome (FAS) and partial FAS, both of which can cause growth problems, central nervous system problems, and characteristic facial features (including small eye openings, flattening of the ridge between the nose and lip, and a thin upper lip), in addition to problems with learning and behavior. People with alcohol-related neurodevelopmental disorder (ARND) don’t have the characteristic facial features or growth deficiency of FAS, but they may have wide-ranging neurocognitive disabilities and problems with behavior and learning. These diagnoses overlap with a newer term—neurobehavioral disorder associated with prenatal alcohol exposure (ND-PAE)—a classification first included in the Diagnostic and Statistical Manual of Mental Disorders (Fifth Edition) as a condition requiring further study (Kable, J. A., et al., Child Psychiatry & Human Development, Vol. 47, No. 2, 2016).
Each of the disorders in the fetal alcohol spectrum can cause problems with self-regulation, executive functioning, social skills, and math skills. These deficits often interfere with children’s performance in school and their ability to make friends. Yet while FASD often causes learning difficulties, the symptoms can be unpredictable. “FASD is the most common preventable cause of intellectual disability in the world. But the majority of alcohol-exposed children have a normal IQ,” Chasnoff said. One notable feature of FASD is a gap between intelligence and adaptive functioning, he added. One of his teen patients, for example, has above average intelligence but is unable to read clocks or count money. “In children affected by alcohol exposure, adaptive functioning is significantly lower than IQ,” he said.
Behavioral problems associated with FASD are common, and often misunderstood, said Petrenko. “So many of the symptoms of FASD can look like intentionally willful or oppositional behavior, when really there are underlying neurodevelopmental explanations,” she said. An accurate diagnosis is the first step toward putting supports in place to address those neurodevelopmental challenges and help people with FASD thrive.
Diagnosis and treatment of FASD
The gold standard for FASD diagnosis is a multidisciplinary evaluation looking at physical features, neurobehavioral impairments, and any known history of prenatal alcohol exposure. The assessment typically involves a variety of specialists such as physicians, speech/language pathologists, psychologists, and geneticists. But those comprehensive evaluations are hard to come by. “There are very few FASD clinics that provide full-service diagnosis,” O’Connor said. “It’s estimated that about only 1% of people with prenatal alcohol exposure can get a diagnosis in that type of situation.”
As a result, many children with FASD are falling through the cracks. Chasnoff and colleagues collected data from 547 foster and adopted children and found that within this group 86.5% of youth with FASD had never been diagnosed or had been misdiagnosed (Pediatrics, Vol. 135, No. 2, 2015). “The great majority of children that are affected by alcohol are misdiagnosed and taking inappropriate medications or receiving ineffective therapy,” Chasnoff said. “FASD should be in the differential diagnosis for any child who presents with behavior problems. And while no single discipline can diagnose FASD, psychologists have a major role to play in the diagnosis.”
Psychologists are also instrumental in designing treatments for children with FASD. To date, only a handful of evidence-based interventions have been developed, each targeting different aspects of FASD. Parents and Children Together (PACT), developed by Chasnoff and colleagues, is a 12-week family intervention that works with children ages 6 to 12 years old and their parents or caregivers to improve self-regulation and executive function. PACT builds on techniques learned from treating traumatic brain injury and sensory processing disorders. The research has found that the intervention improves executive functioning and emotional problem-solving in children with FAS and ARND (Wells, A. M., et al., American Journal of Occupational Therapy, Vol. 66, No. 1, 2012).
The Math Interactive Learning Experience (MILE) program, developed by clinical psychologist Claire Coles, PhD, at Emory University, is a tutoring intervention designed to improve math knowledge and skills, a common area of struggle for children with FASD. A study showed that the 6-week intervention improved both math skills and behavior in alcohol-affected children ages 3 to 10 (Journal of Developmental & Behavioral Pediatrics, Vol. 30, No. 1, 2009).
Children with FASD often have trouble learning social skills as well. The Good Buddies program, developed by O’Connor and colleagues, is designed to teach those skills in a group format over 12 weeks to children ages 6 to 12. The program is derived from an evidence-based treatment for improving children’s friendships, adapted for the specific behavioral and cognitive deficits common in children with FASD (Laugeson, E. A., et al., Child and Family Behavior Therapy, Vol. 29, No. 3, 2007).
The Families Moving Forward Program, created by Heather Carmichael Olson, PhD, and colleagues at Seattle Children’s Research Institute, provides support for families of children with FASD and significant behavioral challenges. The program targets caregivers rather than children themselves and typically lasts about 9 months, in person or by telehealth. Studies have shown the efficacy of the program (Bertrand, J., Research in Developmental Disabilities, Vol. 30, No. 5, 2009), which is now used in multiple states and Canada. Petrenko is collaborating with Olson and colleagues to develop a mobile app, Families Moving Forward (FMF) Connect, to help more families access resources and support (JMIR Formative Research, Vol. 5, No. 12, 2021). The researchers are also adapting the program for children from birth to age 3.
With the right tools, children and adults with FASD can lead successful lives. “The biggest thing we’ve learned is the idea of reframing—looking at behavioral symptoms in a new way,” Petrenko said. Instead of treating a child as oppositional, for instance, reframing helps providers and parents understand that the child may be unable to do what they’re asked because of working memory deficits or other cognitive impairments. “By reframing these interpretations, you can put supports in place to help people be more successful,” she said.
Preventing FASD, attacking stigma
Efforts are also underway to prevent babies from being born with FASD. The Centers for Disease Control and Prevention promotes two strategies to reduce alcohol-exposed pregnancies. CHOICES is an evidence-based program that helps women make decisions around drinking and contraception (Floyd, R. L., et al., American Journal of Preventive Medicine, Vol. 32, No. 1, 2007). The other strategy, alcohol screening and brief intervention (SBI), is a preventive service that involves screening questions about drinking patterns, a short conversation with patients who drink more than recommended amounts, and referral to treatment when appropriate (Planning and Implementing Screening and Brief Intervention for Risky Alcohol Use [PDF, 2.11MB], Centers for Disease Control and Prevention, 2014). “These interventions could easily be incorporated as part of a psychologist’s practice,” O’Connor said. (See more on brief screening interventions.)
Other efforts are underway to reduce the number of children born with FASD. Stoner directs the Washington State Parent-Child Assistance Program (PCAP), a 3-year intensive case management program for mothers who have used alcohol or drugs during pregnancy. PCAP works with pregnant women to stop drinking and also continues to provide support after they give birth. The program connects mothers to social and health services to reduce the likelihood that their future children will be exposed to alcohol or drugs prenatally by reducing substance use or deferring pregnancy. PCAP has 15 sites in Washington covering 19 counties and 90% of the state population, Stoner said. While the program has had success, it was developed several decades ago, and its wide dissemination across the state makes it difficult to do a modern trial to evaluate its effectiveness. To develop that evidence base, Stoner and colleagues have launched a randomized controlled trial in Oklahoma, where they will compare outcomes for women in PCAP with those who receive services as usual.
While education and awareness of FASD have increased among physicians and mental health providers, many are still reluctant to speak with pregnant women about substance use, O’Connor said. Clinical psychologists can and should raise the topic with women in their care who are or might become pregnant. “Prevention can begin in the therapy room,” Stoner said. But it’s important to ask a woman about pregnancy and substance use in ways that encourage honesty and reduce stigma, O’Connor added. “So, for example, instead of asking, ‘Did you drink during pregnancy?’, it’s better to ask, ‘How often did you drink before you found out you were pregnant? And how much did you drink after?’” she said.
While careful conversation can help, stigma continues to be a challenge. Discomfort around the subject often prevents medical providers from asking women about alcohol use during pregnancy at all. Stigma also prevents women from seeking help for alcohol dependence and may prevent them from pursuing a FASD diagnosis for their child. Addressing negative perceptions about alcohol use during pregnancy is an important step toward reducing rates of FASD and improving lives for people with these conditions, Petrenko said. “People with FASD and their families are capable. They can thrive if we recognize their strengths and provide appropriate services and supports.
Source:https://www.apa.org/monitor/2022/07/news-fetal-alcohol-syndrome

3 big factors that drive resident physician burnout

Jennifer Lubell Contributing News Writer-After surveying more than 20,000 physicians and other health professionals across the country, Mark Linzer, MD, has learned a great deal about the drivers of burnout—and possible remedies.
Physician burnout demands urgent action
The AMA is leading the national effort to solve the growing physician burnout crisis. We’re working to eliminate the dysfunction in health care by removing the obstacles and burdens that interfere with patient care.
“Feeling valued was a big mitigator, with burnout rates 30% lower if present. Teamwork was also a big mitigator, while work overload and fast-paced environments were key aggravators,” said Dr. Linzer. He was lead author of the study reporting on these findings that was published in JAMA Health Forum™.
Burnout is real. Rates skyrocketed at the end of 2021 to over 60%, noted Dr. Linzer, who is vice-chief of medicine at Hennepin Healthcare in Minneapolis and also directs the Institute for Professional Worklife there. Making changes at the residency training level is an important strategy for tackling burnout, he advised.
Research by Dr. Linzer and colleagues has revealed “several strong correlates of work conditions with resident burnout, which means there are many ways that programs can address this,” he said. Work-life factors such as teamwork, control of workload, fast-paced, chaotic environments, and time pressure can all affect burnout.
Researchers also identified three resident-specific items contributing to burnout:
- Sleep impairment.
- Program recognition of the resident.
- Interruptions.
“One of our key findings is that work overload and sleep matter, even in the era of duty-hour restrictions,” noted Dr. Linzer.
Residency programs that take physicians’ well-being seriously are more attractive to residency applicants, he stressed. In an episode of “AMA Update,” Dr. Linzer discussed the innovative tool he uses to analyze resident burnout and specific actions residency program and health system leaders can take to increase well-being.
Mini Z research
Dr. Linzer developed the Mini Z measurement instrument, a tool that efficiently measures burnout. It takes two minutes to complete, reducing a six-page survey to a single page.
“Recent studies show it performs very well in measuring in terms of reliability and validity,” said Dr. Linzer. Mini Z versions exist for physicians, residents, nurses, leaders and other clinical staff.
Mini Z core items include three outcomes—satisfaction, stress and burnout, and seven predictors, including the main burnout causes of time pressure. There’s also the three C’s—control, chaos and culture—such as values alignment with leaders.
Translated into several languages, it’s used throughout the world.
Reducing physician burnout is a critical component of the AMA Recovery Plan for America’s Physicians.
Far too many American physicians experience burnout. That’s why the AMA develops resources that prioritize well-being and highlight workflow changes so physicians can focus on what matters—patient care.
Innovations to promote well-being
Evidence-based program interventions usually work best at mitigating and prevent resident physician burnout, advised Dr. Linzer. These may include jeopardy coverage for essential life events, a newsletter celebrating resident achievements, removal of after-hours consult pager call, an extra day off for senior residents on the wards, and care packages distributed through night teams.
“Faculty being on the alert for adverse work environments, such as excess admissions and inability for residents to unplug from the work environment and head home, or in people being distanced on rounds—so they’re not really connecting—might prompt faculty to go deeper and discuss with the resident or program director if they can help,” Dr. Linzer said.
Residency program leaders should also involve residents in data review and interventions. “This is a team effort,” he said. “Let the team guide what needs to be changed and where to go and then let you know if you got there.”
Learn more with the AMA STEPS Forward® toolkit, “Resident and Fellow Burnout: Create a Holistic, Supportive Culture of Well-Being.”

Experts weigh in on the joys and woes of virtual nursing

PULSE By Hunter Boyce, The Atlanta Journal-Constitution Feb 22, 2023
Telehealth has reached new heights in popularity following a workforce-crippling pandemic. That being said, not all healthcare heroes are behind the concept of virtual nursing.
According to a survey by NSI Nursing Solutions, registered nurse turnover stood at roughly 27% in 2022. Meanwhile, RN vacancy rates were at 17.1%. According to Medpage Today, all of those nursing vacancies have hit the healthcare industry with a growing knowledge gap. Virtual nursing is partially designed to close that gap.
New nurses are “scared to death” of making a mistake when they first come onto the floor, Wendy Deibert, MBA, BSN, senior vice president of clinical solutions for Caregility, told Medpage Today.
“They’re thrown into a world… with not a lot of experience behind them,” she said. “So having a button on the wall where you can push… at a moment’s notice and get a nurse in that room to assist (is a huge help).
“I can zoom in to [see] exactly what they’re doing and give direction and support, so that they don’t feel like they’re out there on a limb by themselves. Not only does that boost their confidence, but it also really stops that turnover, because if they get too scared and do not feel supported, they’re not going to stay there.”
Steve Polega, BSN, RN, chief nursing officer of University of Michigan Health-West, however, believes utilizing virtual nursing is a lost cause.
“As a nurse of 25 years, I believe that nursing is a calling and a gift,” he told Becker’s Hospital Review. “It is a huge responsibility to be trusted by our patients and families to be the eyes, ears and caring hands at the bedside. Nursing is all about connecting with people. To earn that trust, I believe that you need to be at the bedside. Nursing is about that kind touch, that smile, those reassuring things that we can do for patients and families.
“It is very challenging to have that real human connection through virtual care. I think we all lose if this trend continues. We have to optimize our technologies to make our nurses more efficient and effective, but at the end of the day, nurses put the humanity into care and need to be present and at the bedside.”
It’s a point that perhaps needs to be put to the test.
Saint Luke’s Health System of Kansas City took advantage of an opportunity to significantly implement virtual nursing in 2019, before the pandemic. The hospital constructed a 33 bed nursing unit at one of its four facilities, utilizing a new care model and workflow.
“It was important that the model had an impact for both the nursing staff and the patient experience,” Jennifer Ball, RN, BSN, MBA, director of virtual care at Saint Luke’s Health System, told the American Nurses Association. “We looked at what could be taken off the plate of the bedside RN and completed by a nurse on camera in the patient room. We included tasks such as admission database, discharge teaching, medication reconciliation, completing procedure checklists, second nurse sign off for meds/skin checks, general education/teaching for the patients, contacting families, answering questions, and the list goes on.
“When these items are completed by the virtual RN, that frees up the time of the bedside RN to have more time to manage physical needs of the patents, answer call lights sooner, and generally have more time with the patients.”
The unit opened in Feb. 2021 and has since experienced several workflow changes. According to Ball, the unit’s operation since its opening has allowed for a few lessons.
“You can never have too much education, training, and information shared,” she said. “Staff have to be flexible, like change, and be willing to try new things. Start your planning early, be wiling to adjust things, and figure out your technology early on. Get everyone involved from the beginning: other disciplines and staff that will be interacting with the new care model.”

PREEMIE FAMILY PARTNERS

A day in the neonatal follow-up clinic

Nov 23, 2022 CanadianPreemies
What to expect and how to prepare for a neonatal follow-up appointment with your baby born preterm.



FOLLOW-UP
All babies, whether born preterm or term, need to have regular visits with a paediatrician for check-ups and immunisations. Preterm babies will probably need to have more regular and thorough follow-up visits beyond what is usually recommended for babies. The purpose of follow-up visits lies in the surveillance of the baby’s progress in growth and development and looking out for potential problems as early as possible.
In general, follow-up visits are scheduled at 4, 8, 12, 18, 24 and 36 weeks corrected age in the baby’s first year, meaning the age if the baby had been born at the expected time.
Usually these visits are there for assessing and tracking the baby’s growth and discussing feedings and sleeping patterns. The developmental level of the baby regarding sensors and the baby’s physical state is evaluated, as well as checks for jaundice are performed. The doctor will also provide the recommended immunisations for the baby. Any questions parents may have about the baby’s health are discussed.
Some countries offer structured preventive early intervention programmes for very preterm infants such as the ToP programme in the Netherlands. It is funded by the Dutch health insurance, consequently every very preterm infant and parent can get this support after discharge. Parents should always take the chance to consult the health care team before going home about specialised care programmes.
Last but not least follow-up practices or clinics are also forums for exchange with doctors and other parents on their baby’s behavior and on recommendations what to do about it.
Parents are often faced with an ‘information flood’ which can be challenging for them to absorb. Information is often new and specific, and parents – commonly worried about their preterm baby – may be overwhelmed.
Tips to help get the most out of follow-up appointments
Starting a file
It can be very helpful to write down the advice given in a file. This will support parents to run a commentary on the baby’s progress which they ca refer to later. In connection with immunisations and vaccinations the GP or paediatrician will record all vaccinations given to the baby in an international immunisation card. It is important and helpful to keep the record for future medical treatment of the baby to track the vaccination history.
Asking questions
Even if parents may suspect their questions to be amateurish, no health care professional will expect parents to understand the various possible health conditions entirely. It is better to ask twice than to leave a visit with uncertainties.
Managing appointments continuously but not too tightly
Sometimes, follow-up appointments for preterm babies can mount up and families may have more than one fixed date in a week. They can take up a lot of time and be very tiring, especially if families have to travel long distances. If it becomes difficult to manage the number of appointments, asking the health visitor to re-organise some of them, if possible, is a reasonable move in order to keep everyday life manageable.
*** The European Foundation for the Care of Newborn Infants (EFCNI) is the first pan-European organisation and network to represent the interests of preterm and newborn infants and their families.
Source:https://www.efcni.org/health-topics/going-home/follow-up/

NICU Follow-up Program – Brigham and Women’s Hospital

May 18, 2022 Brigham And Women’s Hospital
The Neonatal Intensive Care Unit (NICU) Follow-up Program at Brigham and Women’s Hospital provides close, frequent monitoring for babies who spent time in the NICU. Care is provided from discharge until kindergarten using a comprehensive, team-based approach to ensure the child is meeting all developmental milestones.


Premature twin separated from his sibling, has only lived in hospital for first three years of life

KMOV St. Louis Mar 26, 2019


March of Dimes 2022 Report Card Shows US Preterm Birth Rate Hits 15-year High Rates Increase for Women of All Races, Earning D+Grade

November 15, 2022
March of Dimes, the nation’s leader in mom and baby health, released its 2022 Report Card today, revealing that the U.S. preterm birth rate increased to 10.5% in 2021 – a significant 4% increase in just one year and the highest recorded rate since 2007.1 Despite reporting a slight decline last year, the preterm birth rate has steadily increased since 2014, earning the country a D+ grade in the Report Card. The data also shows persistent racial disparities across maternal and infant health measures that were compounded by the COVID-19 pandemic, making the U.S. among the most dangerous developed nations for childbirth.
The report shows that the number of preterm births increased from 364,487 to 383,082 for women of all races. Black and Native American women are 62% more likely to have a preterm birth and their babies are twice as likely to die as compared to White women. In 2021, preterm birth rates for Black mothers increased from 14.4% to 14.7% and increased from 11.6% to 12.3% for Native American/Alaskan Native mothers. What’s more, while Asian women saw a 3% decline in births, they had the largest increase (8%) in preterm births compared to all other women.
Several factors may contribute to the high rate of preterm births, including inadequate prenatal care and preexisting maternal health conditions such as hypertension and diabetes. Over 21.1% of Black women and 26.8% of American Indian/Alaskan Native women in the U.S. do not receive adequate prenatal care. The pandemic has further exacerbated the struggle for parents to access maternal care from hospitals and other prenatal providers.
“This year’s report sheds new light on the devastating consequences of the pandemic for moms and babies in our country,” said Stacey D. Stewart, President and CEO of March of Dimes. “While fewer babies are dying, more of them are being born too sick and too soon which can lead to lifelong health problems. Pregnant women with COVID have a 40% higher risk of preterm birth and we know more women are starting their pregnancies with chronic health conditions which can further increase their risk of complications. It’s clear that we’re at a critical moment in our country and that’s why we’re urging policymakers to act now to advance legislation that will measurably improve the health of moms and babies.”
The report also reveals that low-risk Cesarean births remain alarmingly high, with the highest rates among Black mothers (31.2%). Overall Cesarean delivery rates increased from 31.8% to 32.1% in 2021 and represent nearly one third of all births. While Cesarean birth is lifesaving in medically necessary situations, this form of delivery is a major surgery and does have immediate and long-term risks. With about eight in 10 maternal deaths now preventable according to the CDC, reducing rates of Cesarean births may reduce adverse maternal health outcomes associated with medically unnecessary Cesarean birth.
“We know that the pandemic impacted the way that providers delivered care. Low staffing, resource issues, and fears around COVID-19 transmission put added pressure on providers to get patients delivered and out of maternity units in a timely fashion, and may have also contributed to increases in use of obstetric interventions such as inductions and Cesareans,” said Dr. Zsakeba Henderson, Senior Vice President and Interim Chief Medical and Health Officer at March of Dimes. “These interventions have also been shown to contribute to the rise in preterm births, especially late preterm births.”
For this reason, March of Dimes is working to reduce adverse outcomes driven by non-medically indicated inductions and Cesareans.
March of Dimes recognizes that the maternal and infant health crisis does not have one root cause or a singular solution. Present day structures and systems rooted in racist, biased and unfair policies and practices over centuries contribute to and magnify racial differences in access to resources, social conditions and opportunities.
To better understand and address the social drivers to healthcare, this year’s report includes the Maternal Vulnerability Index (MVI) – a new measure of the contextual, clinical, and social determinants of health that impact pregnant people and their babies. Developed by Surgo Ventures, the MVI is the first county-level, national-scale, open-source tool to identify where and why moms in the U.S. are vulnerable to poor health outcomes. It explores 43 indicators across six themes, including reproductive health care, physical health, mental health and substance use, general health care, socioeconomic determinants, and environmental factors. The MVI shows that while some parts of the country are more vulnerable, 4 out of 5 counties have some aspect of maternal health that can be improved. Black women in the lowest vulnerability counties are still at higher risk of death and poor outcomes than White women living in the highest vulnerability counties.
Supplemental Report Cards also provide an in-depth analysis of the national and state maternal and infant health data found in the report. New this year, the reports include a summary of March of Dimes programmatic initiatives and legislative advocacy efforts in each state.
2022 March of Dimes Preterm Birth Grades
Each year, the March of Dimes releases its Report Card with grades for individual states, Washington, D.C., Puerto Rico and the 100 cities with the greatest number of births. Between 2020 and 2021, 45 states, Washington D.C. and Puerto Rico experienced an increase in preterm birth rates.
- 9 states and Puerto Rico earned an “F” (Alabama, Arkansas, Georgia, Kentucky, Louisiana, Mississippi, Oklahoma, South Carolina, West Virginia)
- 4 states earned a “D-” (Missouri, Nevada, Tennessee, Texas)
- 6 states earned a “D” (North Carolina, Nebraska, Florida, Indiana, Delaware, Wyoming)
- 5 states earned a “D+” (Ohio, Illinois, Michigan, Maryland, South Dakota)
- 2 states and Washington D.C. earned a “C-“(Hawaii, Alaska)
- 11 states earned a “C” (Arizona, Colorado, Iowa, Kansas, Montana, New Mexico, New York, Pennsylvania, Utah, Virginia, Wisconsin)
- 5 states earned a “C+” (North Dakota, Connecticut, Maine, Minnesota, Rhode Island)
- 4 states earned a “B-” (New Jersey, Massachusetts, California, Idaho,)
- 2 states earned a “B” (Washington, Oregon)
- 1 state earned a “B+” (New Hampshire)
- 1 state earned an “A-” (Vermont)
Actions to Address the Crisis
Alongside the release of the report, March of Dimes is delivering the Mamagenda for #BlanketChange, an emergency call-to-action to Congress to improve the health of moms and babies. The Mamagenda calls for immediate action to advance policies that support equity, access and prevention, advocating for the enactment of the Black Maternal Health Act of 2021 (H.R. 959/S. 346) and the Pregnant Workers Fairness Act (H.R. 1065/S.1486) to help prevent racial and ethnic discrimination in maternity care, expand access to midwifery care, provide reimbursements for doula support, and more. It calls for adopting Medicaid expansion and permanently extending Medicaid postpartum coverage to 12 months as authorized under the American Rescue Plan Act. Additionally, the Mamagenda calls for funding for Maternal Mortality Review Committees and Perinatal Quality Collaboratives that work to improve data collection for maternal deaths and make improvements in quality of care and maternal and infant health outcomes.
Visit BlanketChange.org to learn more and join the growing number of partners committed to improving maternity care for all.

INNOVATIONS

Babies born in rural settings are more likely to experience trauma during birth, and one way Mayo Clinic is addressing this
By Elizabeth Zimmermann – January 25, 2022
Birth trauma rates are one of the measures of hospital quality used by the Joint Commission. Recent Mayo Clinic-led research, published in the Maternal and Child Health Journal, shows that babies born at rural hospitals are more likely to experience a birth-related injury than those born in urban hospitals.
This disparity is of concern to researchers and clinicians.
To address gaps and disparities in care, the Mayo Clinic Robert D. and Patricia E. Kern Center for the Science of Health Care Delivery works with the medical practice to investigate factors that contribute to high quality, high value care.
“In order to provide care that meets the needs of patients and the overall population, there is a need to understand current outcomes, in the context of current care settings and processes,” says Aaron Spaulding, Ph.D., a health services researcher at Mayo Clinic in Jacksonville, Florida, and the study’s senior author.
This is not Dr. Spaulding’s first study into the disparities of care and outcomes that are multi-faceted and not easily assessed. Within the Mayo Clinic Robert D. and Patricia E. Kern Center for the Science of Health Care Delivery, he has led several multiinstitutional collaborations investigating differences in hospital quality outcomes between geographical settings, including the current study.
“Our work in this area seeks to understand better how communities in which hospitals reside influence hospital outcomes and vice versa,” he says. “We are led by the belief that many policies attempt to use a one-size-fits-all mentality which may be inappropriate and may punish or reward hospitals based on aspects they have little control over.”
Dr. Spaulding and his team hope that as they gain a better understanding of the association between communities and their hospitals, they will find better opportunities for policy or practice interventions that can make a difference.
Babies in distress
In the current study, Dr. Spaulding, along with Hanadi Hamadi, Ph.D.; Jing Xu, Ph.D.; and Farouk Smith all of the University of North Florida, Jacksonville; and Aurora Tafili, University of Alabama at Birmingham; used Florida hospitals’ inpatient data from 2013 to 2018. Originally collected by the Florida Agency for Health Care Administration, the study data included information from 125 inpatient hospitals across the state. It included information for 1,192,336 singleton births and noted up to 31 diagnoses present on admission, as well as up to 31 injury-related diagnoses for the births.
“The most notable finding of the study is that no matter your race, a rural location was associated with an increased odds of birth trauma compared to an urban location,” says Dr. Spaulding.
His team is especially concerned about people of Hispanic ethnicity receiving care at a rural location, he continues, since the greatest birth trauma risk was among rural Hispanic or Latino babies.
The dataset they used only included Florida, however many states make this type of data available, which could be used for a similar analysis. It would not be unreasonable to assume that many states would show disparities between urban and rural outcomes much like those the researchers found in Florida, he says.
Teleneonatology consult allows specialist to see what the local care team is seeing, and to direct lifesaving care for infants in distress.
A possible solution to rural health care disparities at birth
Telemedicine has taken hold as a viable means by which people can access care not available in their geographic area. Mayo Clinic has been steadily implementing and evaluating a wide range of solutions to connect with patients wherever they are, and whenever they need that connection.
For babies born in rural settings or even urban hospitals with no access to neonatologists — pediatricians specializing in medical care for newborns (neonates) — Mayo’s Teleneonatology Program may bridge an essential gap, leading to better outcomes following birth-related traumas like those noted in Dr. Spaulding’s study.
“With this technology, we can be at the bedside of any newborn in need of critical care,” says Jennifer Fang, M.D., medical director of Mayo Clinic’s Teleneonatology Program.
In another recent publication, Dr. Fang describes how she and her colleagues are able to use telemedicine to remotely respond to newborn emergencies. In the paper, she notes the significant improvements in outcomes since teleneonatology consultations were integrated into the family birth centers and emergency departments of nineteen participating community hospitals. These include advancements in quality, safety and provider experience.
During a teleneonatology consult, a neonatologist at Mayo Clinic in Rochester, Minnesota, connects with the local care team in real time, via a telemedicine platform incorporating high resolution, secure audio and video capabilities.
“We are able to see and assess the infant, and guide the local doctors and nurses through resuscitation, including positive pressure ventilation, advanced airway placement and umbilical catheter placement, when needed” says Dr. Fang.
“As one of the acute care telemedicine services developed at Mayo Clinic, we were looking for a way to help more babies – even before they arrive to the neonatal ICU,” she says.
“Before our teleneonatology program we would try to help via a phone call with the care team during a particularly complex delivery. But nothing compares to being able to visualize the baby, to see what the local team is seeing, and how the newborn responds in the minutes after birth and following interventions.” Says Dr. Fang.
Other research has shown that approximately 10% of newborns require breathing assistance after delivery, and one in 1,000 require extensive resuscitation. These babies are much more likely to die in when delivered in hospitals with lower levels of neonatal care. Mayo’s Teleneonatology Program aims to reduce that risk.
In Dr. Fang’s paper, she reviews some of Mayo’s program results, including:
- Substantially higher quality resuscitation for infants whose care team used the service.
- Safer care — as demonstrated by significant reduction in birth injury cases.
- Willingness to use the capability is good. In fact, 99% of providers would use teleneonatology support again — and recommend it to their colleagues.
Mayo’s various telemedicine capabilities are enabled by Mayo Clinic’s Center for Digital Health. Much of the research validating and evaluating new telemedicine and remote care capabilities is done in collaboration with the Mayo Clinic Kern Center for the Science of Health Care Delivery.
Next steps for researchers
Dr. Spaulding’s team continues to work on topics assessing disparities, geographic location and care outcomes. Also in an effort to understand factors that contribute to healthier infants, they are assessing the value of designation under the Baby-Friendly Hospital Initiative. They hope to determine which hospital and community characteristics are associated with hospital attainment of the designation.
On a broader scope, he and his colleagues seek to better understand the effect of community characteristics and health care outcomes. For example, his team is evaluating the presence of Magnet-designated hospitals and differences in associated health care outcomes between Magnet and non-Magnet-designated hospitals. (Read a related publication, “The influence of community health on hospitals attainment of Magnet designation: Implications for policy and practice.”)
“We hope to develop further our understandings of how community characteristics influence health outcomes and how hospital characteristics affect community health,” says Dr. Spaulding.
Dr. Fang agrees that more research will be helpful for her program in particular.
“If we could get this program into every rural setting, I am confident we would see positive health outcomes for babies,” she says. “Research can help us determine costs and savings (a cost analysis of teleneonatology performed by the Division of Neonatal Medicine and the Kern Center is currently under review for publication), as well as quantifiable public health outcomes that can help shift perceptions among the people and agencies who oversee policies, payment, and care offerings at local, regional, state and national levels.”
In general Drs. Fang and Spaulding both agree that it is high time the health care community pays attention to diversity and inclusion research and the associated attempts to improve care for all. This research sheds light on the importance of the community in which one lives, which impacts health from the cradle to the grave.
“We hope that our research can help further clarify areas of needed policy and practice intervention,” concludes Dr. Spaulding. “Improved measurement of disparities and comparisons between communities and geographic locations will provide us with better tools to fight unequal access to quality care.”
Implementation of A Neurodevelopmental Care Bundle to Promote Optimal Brain Development in the Premature Infant
Author: Pamela S Hackman, MSN, RNC-NIC, C_ELBW Registered Nurse Hershey Medical Center Children’s Hospital, Hershey PA 629 Thoreau Drive 7173301589 phackma@hotmail.com
Background and Purpose: When an infant is born prematurely, the external environment, routine or emergent nursing care actions performed on the infant can be detrimental. Neonatal nurses are keenly aware the premature infant is at risk for developing behavioral, cognitive, and physical impairments which can be short term or last a lifetime. The purpose of a neuroprotective care bundle is two-fold: First, for nurses, the bundle optimizes the health and well-being of the infant by incorporating seven core measures: healing environment, partnering with families, positioning, and handling, safeguarding sleep, minimizing stress and pain, protecting skin, and optimizing nutrition. Second, for families, therapeutic touch, and skin-to-skin contact cultivates positive neurodevelopmental outcomes, nurturing and health for the infant as well as enhances the bonding experience for the family. Comprehensive, evidence-based research was conducted looking at the role of developmental care and prematurity and how it can correlate to a healthy environment for the premature infant. Result of that research indicates that decreasing negative effects of extrauterine life, decreasing touch times, and implementing a Neuroprotective care bundle in the neonatal intensive care unit can be modified to simulate an intrauterine environment, thereby promoting optimal brain development and outcomes for that infant.
Materials and Methodology: A quantitative research study was conducted in a level 4 neonatal intensive care unit with an average admission rate between 350-400 infants per year, with approximately 120 of those infants are born prematurely. Research was conducted over a twelve-week period. Eighteen premature infants 23-32 weeks gestation were tracked for the first 7 days of life.
A Pareto chart was developed. Information on the chart included: birthweight, and gestational age. The chart was divided into 4-hour increments for a 24-hour (1day total). A list of variables disturbances to the infant included such interventions as opening the top of the isolette for CXR, or other medical test, opening the port holes to the isolette for attaining vital signs including blood pressure, diaper change, repositioning, suctioning, heel stick for blood, parental interaction with infant, answering an apnea, bradycardia, or desaturation alarm, consoling a crying infant, and assessment by medical team. The goal of the project was for the nurse to check off each intervention during an identified time slot. Data was collected for 7 days.
At the end of twelve weeks, each variable in the time interval and tic mark for that time was tabulated. Then all interventions were added together for each day. To find out the average number of times an infant was disturbed, the total number of disturbances per day divided by 7 for the total study period was identified. This information indicated the number of times in a day that an infant was disturbed. Further calculation was done to figure out the number of times per day the infant was disturbed by dividing total number of interventions per day by 24 (hours in a day).
Results: Main outcome results indicated an infant was disturbed between 89 to 242 times during the first week of like. Further breakdown indicated that infants were disturbed 3.7 to 10.1 times per hour. Barriers recognized when research study complete included: staff unaware of study so did not complete project, despite education and communication to all staff members. Multiple shifts did not have documentation complete. Documentation of tic mark for variable but no tic mark for opening port holes (assumption made here). No report of position change. No documentation noted on one patient for one shift. One patient did not have documentation for 2.5 days. Not all activities/interventions were captured. Too busy/ high acuity/ did not understand project request. Multiple pts/activities due at the same time. Totally dependent on RN to document data. Some variables were documented but no documentation for opening the port holes or popping the top of isolette that needed to happen first before taking care of the infant (assumption made here when looking at the intervention completed). Despite interventions being missed in the total tabulation of disturbances to the infant, the study was an eye-opening experience for the nurse to see the total number of times an infant is disturbed per day and per hour. The number of disturbances to the premature infant is detrimental to their health and something that is not often thought about when caring for the infant. Based on the limited results of this study, the intensive care unit in which this study was conducted is currently looking at interventions that promote the developing behavioral, cognitive, and physical needs of the premature infant by instituting specific touch times with infant that correlate with the infant’s wake cycle, implementation of a neurodevelopmental care bundle and promoting a family centered approach to care. To assimilate the intrauterine environment a neurodevelopmental care bundle ought to be utilized.
Conclusion: A family- care, neuroprotective and developmentally supportive care approach, in conjunction with standard of care practices, promote brain development and a healthy environment. The implementation of a neurodevelopmental care bundle provides an opportunity to promote optimal brain development as the infant grows in the intensive care, thereby, fostering a positive experience for the family, decreasing length of stay, decreasing hospital cost, and improving medical outcomes.
Learning Objectives: At the end of this presentation the learner will be able to:
1. Identify the how the implementation of a neurodevelopmental care bundle promotes the developing behavioral, cognitive, and physical aspects of the premature infant.
2. Identify external environmental factors that are detrimental to the premature infant and how the intrauterine environment can be assimilated in the external environment.
3. Identify the positive outcomes of promoting a neurodevelopmental care bundle.
Source:http://neonatologytoday.net/newsletters/nt-jan23.pdf
Mayo Clinic Teleneonatology Program: Simulated Teleneonatology Consult

Mayo Clinic Jun 14, 2017 Mayo Clinic



The Power of Pets Health Benefits of Human-Animal Interactions

Nothing compares to the joy of coming home to a loyal companion. The unconditional love of a pet can do more than keep you company. Pets may also decrease stress, improve heart health, and even help children with their emotional and social skills.
An estimated 68% of U.S. households have a pet. But who benefits from an animal? And which type of pet brings health benefits?
Over the past 10 years, NIH has partnered with the Mars Corporation’s WALTHAM Centre for Pet Nutrition to answer questions like these by funding research studies.
Scientists are looking at what the potential physical and mental health benefits are for different animals—from fish to guinea pigs to dogs and cats.
Possible Health Effects
Research on human-animal interactions is still relatively new. Some studies have shown positive health effects, but the results have been mixed.
Interacting with animals has been shown to decrease levels of cortisol (a stress-related hormone) and lower blood pressure. Other studies have found that animals can reduce loneliness, increase feelings of social support, and boost your mood.
The NIH/Mars Partnership is funding a range of studies focused on the relationships we have with animals. For example, researchers are looking into how animals might influence child development. They’re studying animal interactions with kids who have autism, attention deficit hyperactivity disorder (ADHD), and other conditions.
“There’s not one answer about how a pet can help somebody with a specific condition,” explains Dr. Layla Esposito, who oversees NIH’s Human-Animal Interaction Research Program. “Is your goal to increase physical activity? Then you might benefit from owning a dog. You have to walk a dog several times a day and you’re going to increase physical activity. If your goal is reducing stress, sometimes watching fish swim can result in a feeling of calmness. So there’s no one type fits all.”
NIH is funding large-scale surveys to find out the range of pets people live with and how their relationships with their pets relate to health.
“We’re trying to tap into the subjective quality of the relationship with the animal—that part of the bond that people feel with animals—and how that translates into some of the health benefits,” explains Dr. James Griffin, a child development expert at NIH.
Animals Helping People
Animals can serve as a source of comfort and support. Therapy dogs are especially good at this. They’re sometimes brought into hospitals or nursing homes to help reduce patients’ stress and anxiety.
“Dogs are very present. If someone is struggling with something, they know how to sit there and be loving,” says Dr. Ann Berger, a physician and researcher at the NIH Clinical Center in Bethesda, Maryland. “Their attention is focused on the person all the time.”
Berger works with people who have cancer and terminal illnesses. She teaches them about mindfulness to help decrease stress and manage pain.
“The foundations of mindfulness include attention, intention, compassion, and awareness,” Berger says. “All of those things are things that animals bring to the table. People kind of have to learn it. Animals do this innately.”
Researchers are studying the safety of bringing animals into hospital settings because animals may expose people to more germs. A current study is looking at the safety of bringing dogs to visit children with cancer, Esposito says. Scientists will be testing the children’s hands to see if there are dangerous levels of germs transferred from the dog after the visit.
Dogs may also aid in the classroom. One study found that dogs can help children with ADHD focus their attention. Researchers enrolled two groups of children diagnosed with ADHD into 12-week group therapy sessions. The first group of kids read to a therapy dog once a week for 30 minutes. The second group read to puppets that looked like dogs.
Kids who read to the real animals showed better social skills and more sharing, cooperation, and volunteering. They also had fewer behavioral problems.
Another study found that children with autism spectrum disorder were calmer while playing with guinea pigs in the classroom. When the children spent 10 minutes in a supervised group playtime with guinea pigs, their anxiety levels dropped. The children also had better social interactions and were more engaged with their peers. The researchers suggest that the animals offered unconditional acceptance, making them a calm comfort to the children.
“Animals can become a way of building a bridge for those social interactions,” Griffin says. He adds that researchers are trying to better understand these effects and who they might help.
Animals may help you in other unexpected ways. A recent study showed that caring for fish helped teens with diabetes better manage their disease. Researchers had a group of teens with type 1 diabetes care for a pet fish twice a day by feeding and checking water levels. The caretaking routine also included changing the tank water each week. This was paired with the children reviewing their blood glucose (blood sugar) logs with parents.
Researchers tracked how consistently these teens checked their blood glucose. Compared with teens who weren’t given a fish to care for, fish-keeping teens were more disciplined about checking their own blood glucose levels, which is essential for maintaining their health.
While pets may bring a wide range of health benefits, an animal may not work for everyone. Recent studies suggest that early exposure to pets may help protect young children from developing allergies and asthma. But for people who are allergic to certain animals, having pets in the home can do more harm than good.
Helping Each Other
Pets also bring new responsibilities. Knowing how to care for and feed an animal is part of owning a pet. NIH/Mars funds studies looking into the effects of human-animal interactions for both the pet and the person.
Remember that animals can feel stressed and fatigued, too. It’s important for kids to be able to recognize signs of stress in their pet and know when not to approach. Animal bites can cause serious harm.
“Dog bite prevention is certainly an issue parents need to consider, especially for young children who don’t always know the boundaries of what’s appropriate to do with a dog,” Esposito explains.
Researchers will continue to explore the many health effects of having a pet. “We’re trying to find out what’s working, what’s not working, and what’s safe—for both the humans and the animals,” Esposito says.
The Power of Pets | NIH News in Health

Dogs or cats with SUPERPOWER?!

Dec 5, 2018 CurioSips
Dogs or cats with SUPERPOWER?! We all have had that one time at least that our pet goes crazy and scratches us for no reason! Or when your cat starts staring at the window but there is nothing there? That is what happens at my house every single day! No matter how exaggerated these things seem, if this happens in your house as well, it might be that your pet is truly haunted, didn’t you think?


Kat’s Update:
When the pandemic hit, I was in the second year towards pursuing my medical education. Due to the impact of the pandemic on medical education and clinical surgery education in particular, I chose to defer and postpone my medical studies.
In order to progress my knowledge, engagement, and expertise in global surgery and the medical community I have continued to participate in ongoing academic and independent research. Over the past two years, I have had the privilege and pleasure of presenting my research at 8 conferences in over 3 countries, expanding my professional network and growing my passion for advocacy and promotion of surgical care globally.
During the past year, I chose to pursue my MSc in London with a focus on global surgery and research pertaining to surgical system strengthening in austere environments. The opportunity to learn from and study alongside my fellow global surgery pathway cohort members and our respective global health cohort has allowed me to build strong relationships and gain close colleagues from over 15 nations.
Perhaps the most impactful aspect of my program was the gift of gaining unimaginably strong friendships with four of my colleagues, each of whom are physicians from different countries (England, Ireland, Colombia, Ethiopia), all of whom embrace career aspirations in various areas of global surgery/medicine including obstetrics, neurosurgery, otolaryngology, and anesthesia. Each of these individuals has inspired me to become more present, gracious, composed, and joyous in my life and interaction with others.
To my brilliant, compassionate, strong, and resilient friends Oscar, Martina, Heaven, and Tina THANK YOU for sharing your wisdom, hopes, dreams, and kind hearts! Your support and friendship have strengthened my ongoing intention towards completing my medical education. I look forward to the day I can join you all in service as a physician.
To my amazing cohort, I am GRATEFUL for the various perspectives, intellect, care, love, joy, passion, fire, fun, and the positive challenges you have each provided us as a whole in order for us all to grow, develop, and strive to become better global citizens.
Beloved Neonatal Womb Warrior Brothers and Sisters! Your unique and personal journeys will create joyful and meaningful opportunities for magnificent manifestations and personal growth. Please take a moment or two to breath, relax, acknowledge, and experience the gratitude you feel towards those in your lives who gift you with their presence and spectacular beingness……
In 2023, I look forward to continued engagement in professional research with the goal of strongly contributing to the mission of those I have the pleasure of working alongside and towards creating a tangible impact in the communities and lives we seek to serve.
Kathy and Kat: Our precious and powerful Neonatal Womb Warrior/Preterm Birth Family! Our hearts are continually vitalized by your powerful presence. Every month you educate, challenge us towards change, surprise, and enchant us through your intellect, humanity, and courage. As we voyage forward into this next year, the seventh year of our Neonatal Womb Warrior collaboration; let us live wholeheartedly, let us remember the moments in life which empower our presence, the people in our lives who light up our world, that we are capable of living our dreams, and that with open hearts we belong to each other!
Let us go forward fully and fiercely, immersed within the journeys of our destinies…….
Pets! They are just full of surprises! The highlighted video shows us a primary example of the kind of lighthearted fun and joy pets bring us each day!
In my experience with our cat, Gannon, he has often taken us off-guard by scattering his numerous toys in odd places and through occasionally pouncing on our feet from underneath a bed as we pass. Perhaps the most fulfilling surprise he has graced us with is his requirement that when we show him affection, we must allow him to give it back (licking/cleaning and gripping our hands, snuggling).
Throughout the years each of our pets has brought us great joy and a sense of belonging in our lives. Pets are not just family; for me they are guardian angels who help me navigate the world and provide opportunities to learn more about myself and my relationships with others. The countless pets in our neighborhood have certainly helped me develop newfound friendships and participate in important, unexpected, and depth-filled conversations with others. There have been a scattering of belly laughs and a few occasional tears, focused on owner love for their pet!
It’s never a dull moment when the pets are front and center. My hope is the comfort, love, and even those pesky and annoying challenges they bring about in our daily lives may help encourage us to send out unconditional love into the world in the ways in which they do every day.
Do you have a pet? What do the pets in your life inspire?
Surf Team Hungary – 1. Rész

Peiman Lotfi Sep 30, 2013
We have chosen a serious challenge for the 2013 surfing season, because this year the first Hungarian surfing team was assembled, which for the first time in history will compete in the European Championship (Eurosurf 2013) held this year in the Azores Islands. Unfortunately, the team was not able to enter the originally planned full team, as some key surfers could not come, especially Miki Rigler, but we still have 4 competitors in the “Open Men” category. By name, András Ajtai, Lotfi Peiman, Dávid Liptay and Krisztián Kövesdán. In the first part, we introduce our players and learn about the history of participation in the European Championship.











































































 From our third floor window, while a very inspired woodpecker hammers our wood/concrete siding in order to mark his territory, we greet you with our love, gratitude, and very best wishes!
From our third floor window, while a very inspired woodpecker hammers our wood/concrete siding in order to mark his territory, we greet you with our love, gratitude, and very best wishes! 

























































 Published on Jan 31, 2019 Physician Mental Health & Suicide
Published on Jan 31, 2019 Physician Mental Health & Suicide



















