

Estonia, officially the Republic of Estonia, is a country in the Baltic region of Northern Europe. It is bordered to the north by the Gulf of Finland across from Finland, to the west by the Baltic Sea across from Sweden, to the south by Latvia, and to the east by Russia. The territory of Estonia consists of the mainland, the larger islands of Saaremaa and Hiiumaa, and over 2,300 other islands and islets on the east coast of the Baltic Sea. Its capital city of Tallinn, along with the city of Tartu, are the country’s two largest urban areas. The Estonian language, of the Finnic family, is the official language and the first language of the majority of nearly 1.4 million people. Estonia is one of the least populous member states of the European Union.
Present-day Estonia has been inhabited since at least 9,000 BC. The medieval indigenous population of Estonia was one of the last pagan civilisations in Europe to adopt Christianity following the Northern Crusades in the 13th century. After centuries of foreign rule by the Teutonic Order, Denmark, Poland, Sweden, and the Russian Empire, a distinct Estonian national identity gained new momentum with the Age of Awakening in the mid-19th century. This culminated in the 1918 Estonian Declaration of Independence. Democratic throughout most of the interwar period, Estonia declared neutrality at the outbreak of World War II, but the country was repeatedly invaded and occupied, and ultimately annexed into the USSR. Throughout the de facto Soviet occupation, from World War II until 1991,] Estonia’s de jure state continuity was preserved by diplomatic representatives and the government-in-exile. Following the 1988–1990 “Singing Revolution” against Soviet rule, full independence was restored on 20 August 1991, and the country joined NATO and the European Union in 2004.
Estonia is a developed country with a high-income, advanced economy, and Eurozone membership. It is a democratic unitary parliamentary republic, with a single-tier local government system consisting of 79 municipalities. Estonia is among the least corrupt countries in the world and ranks very high in international rankings for education, human development, press freedom, online public services, and the prevalence of technology companies.
Healthcare in Estonia is supervised by the Ministry of Social Affairs and funded by general taxation through the National Health Service.
The service is administered by the Estonian Health Insurance Fund (EHIF). An insured person must be either a permanent resident or a legal resident who pays the social tax. All health care providers in Estonia are required to submit the health information of their patients to the digital health information system.
Estonia’s health care system is based on compulsory insurance based on solidarity funding and on universal access to services provided by private service providers. All providers of health services are autonomous businesses governed by private law. The single buyer and payment method is the Estonian Health Insurance Fund (Eesti Haigekassa), which pays all contracted providers. The majority of general practitioners work for themselves, privately owned businesses, or local governments. In Estonia, the majority of hospitals are either foundations created by the government, municipalities, or other public organizations, or limited businesses owned by the local government. If the hospital has a contract with the Fund, the Estonian Health Insurance Fund will also pay for necessary treatments received in a private hospital. If no agreement is reached, private medical care is not reimbursed.
Source: https://en.wikipedia.org/wiki/Estonia
- GLOBAL PRETERM BIRTH RATES – Estonia
- Estimated # of preterm births: 6 per 100 live births
- (Global Average: 10.6)
- Source- https://worldpopulationreview.com/country-rankings/preterm-birth-rates-by-country



https://www.frontiersin.org/journals/pediatrics/articles/10.3389/fped.2025.1565897/full



Children born with single‑ventricle heart disease, a rare and serious heart defect, often undergo multiple surgeries in their first years of life. A new study shows the challenges for these children can last well into adolescence and sometimes throughout their lives.
Researchers from Duke Health and the Pediatric Heart Network followed 549 children with single ventricle heart disease for 16 years and found that 87% either died or developed a major health problem over time. Only 12% reached adolescence without a significant complication.
These findings, published in the Journal of the American College of Cardiology March 11, give families a clearer picture of what to expect and offer the medical community new direction for improving care.
“Now that we understand these trajectories and the risk factors for suboptimal outcomes, we can counsel families more clearly and tailor therapies to each child.”
Kevin D. Hill, M.D., corresponding author, chief of division of pediatric cardiology and professor, Department of Pediatrics, Duke University School of Medicine
The study introduces a new way of evaluating long term health called a global rank score. It’s a measure developed with input from families that captures what matters most to them, including daily functioning, quality of life, and heart performance.
One of the most important findings for families is that most children will continue to need specialized care long after surgery. The study found high rates of adaptive behavior delays, quality of life challenges, and reduced heart function in many patients, often emerging gradually as they grow.
Researchers also found that premature babies faced the steepest uphill climb. More than 60% of premature infants in the study died, and only 3% reached adolescence without major problems. The data suggests that supporting healthy pregnancies and preventing preterm birth could make a meaningful difference.
For clinicians, the study provides new clues that may help tailor early surgical decisions. Although the two main shunt types used in infancy showed similar long-term outcomes overall, babies who had moderate or severe tricuspid valve leakage before surgery did worse with the right ventricle shunt, a finding that could guide future decision making.
Just as importantly, the study revealed substantial differences among hospitals. Some centers had lower early mortality, while others excelled in long term follow up and rehabilitation. This variation highlights opportunities to share best practices and improve care no matter where a child is treated.
“These children need lifelong comprehensive care, including medical, developmental, and school support,” Hill said. “It’s not like they get surgery and are ‘fixed.’ Ongoing, coordinated services are essential to help them thrive.”
The study emphasizes that children with single ventricle heart disease often face challenges in multiple areas of life. The research team hopes these findings will help families feel better prepared, while giving healthcare teams a roadmap for improving outcomes through physical therapy, mental health support, school accommodations, exercise programs, and strong long-term monitoring.
In addition to Hill, study authors include, Lillian Kang, Qiao Wang, Jane W Newburger, J. William Gaynor, Kristin M. Burns, Richard Williams, Felicia Trachtenberg, Brian W. McCrindle, Eric M. Graham, Christian Pizarro, Awais Ashfaq, Andrew Souza, James A. Kuo, Dawn Ilardi, Kurt R. Schumacher, Hwanhee Hong, and Caren S. Goldberg.





Abstract: Premature birth shifts late fetal organ maturation into the extrauterine environment, creating overlapping biologic windows during which organs remain structurally immature yet actively developing and highly sensitive to neonatal exposures. Because organ systems mature in parallel, vulnerability evolves across postmenstrual age (PMA) rather than discrete gestational stages. Prematurity unfolds as a developmental continuum in which the dominant focus of neonatal care shifts with maturation. This review integrates developmental physiology with evolving care priorities across four maturational phases: early transition (lung– cardiovascular, brain, kidney), interface (skin, gut–immune, retina), growth–accretion (bone–mineral, hematologic, somatic growth), and near-term neurointegrative maturation. Interpreting prematurity through these sequential biologic windows clarifies the timing of prematurity-related disorders and provides a physiologic rationale for neonatal monitoring and management across the course of prematurity.
Introduction: Late gestation is characterized by coordinated maturation across organ systems, including structural growth, angiogenesis, epithelial barrier formation, mineral accretion, and physiologic integration. Preterm birth interrupts this synchronized developmental program and exposes organs to extra-uterine oxygen tension, fluctuating perfusion, artificial nutrition, microbial colonization, and environmental stress while maturation remains incomplete. The neonatal course of prematurity, therefore, reflects shifting biologic windows during which specific systems are actively developing and particularly vulnerable to perturbation.
Because fetal organ maturation proceeds in parallel rather than sequence, vulnerability windows overlap broadly across PMA. For clinical interpretation, prematurity can be conceptualized as progression across phases in which particular organ systems dominate vulnerability while earlier susceptibilities recede.
Importantly, this maturation also corresponds to a developmental shift in neonatal care priorities that parallels changes in organ vulnerability across PMA. This progression of biologic windows across prematurity is outlined below.
Developmental Progression of Care Focus Across Prematurity:
Prematurity unfolds as a continuum in which the dominant determinants of physiologic stability shift with maturation. Immediately after birth, survival depends on fragile organs still constructing the internal physiologic environment. As cardiopulmonary stability improves, organs mediating interaction with the extra-uterine world become the principal sites of vulnerability. With the maturation of these interfaces, development becomes dominated by nutrient-dependent tissue accretion. Although nutrient provision is required throughout prematurity, this stage reflects the period when accretion demand becomes the principal determinant of vulnerability. Near-term equivalent age: maturation culminates in the integration of previously developing systems into coordinated whole-organism function.
Accordingly, neonatal care priorities evolve in parallel with these biologic transitions:
internal stabilization → environmental interface → growth accretion → neurointegrative function
This sequence reflects the organs most actively developing at each PMA and most susceptible to environmental disruption. As maturation proceeds, earlier vulnerabilities recede while new developmental demands emerge, producing the characteristic temporal pattern of prematurity-related disorders and the need for clinical monitoring across the neonatal course.
PHASE 1—EARLY TRANSITION:
Vulnerable Organ Stabilization (Birth to ~30 wk PMA):
Dominant systems: Lung–Cardiovascular, Brain, Kidney:
Immediately after a very preterm birth, survival depends on organs still undergoing fundamental structural and regulatory assembly. The immature lung–cardiovascular system determines oxygenation and systemic perfusion; the developing brainstem governs respiratory rhythm, autonomic tone, thermoregulation, and postural organization; and the immature kidney regulates electrolyte and metabolic homeostasis. Instability within these systems propagates system-wide physiologic variability affecting all organs. Care during this phase focuses on stabilizing these vulnerable transitional organs that establish the internal environment for subsequent development
Lung and Cardiovascular System:
Between approximately 22–28 weeks of gestation, the lung transitions from canalicular to early saccular architecture, with active formation of distal airspaces and pulmonary capillary networks. Pulmonary vascular resistance remains elevated, and the circulatory transition remains incomplete. Exposure to mechanical ventilation, oxygen variability, and inflammation during this stage disrupts septation and capillary alignment. Because rapid alveolarization occurs later (32–36 wk PMA), structural injury sustained during early transition manifests weeks later as evolving bronchopulmonary dysplasia.
Pulmonary immaturity produces intermittent hypoxemia and carbon-dioxide variability that propagate systemic perfusion instability affecting cerebral, renal, and intestinal circulation. Immature ductal smooth muscle contributes to the persistence of the ductus arteriosus. As pulmonary vascular resistance falls toward term PMA, oxygenation stabilizes and hemodynamic variability declines.
Noninvasive distending pressure is commonly continued into late prematurity to support alveolar stability during the transition from saccular to early alveolar development. Although oxygen requirements and work of breathing may improve before term equivalent age, the preterm lung remains prone to derecruitment due to a compliant chest wall, limited surfactant reserve, and an evolving distal airspace-capillary architecture. Many infants remain on nasal CPAP until approximately 34 weeks PMA to preserve functional residual capacity and reduce cyclic atelectasis while pulmonary mechanics and distal airspace maturation continue.
Brain and Neuroregulatory Maturation:
Between approximately 24–32 weeks of gestation, brainstem and hypothalamic networks regulating respiratory rhythm, arousal, autonomic tone, thermoregulation, and postural control remain structurally and functionally immature. Limited central respiratory drive, impaired chemoreceptor responsiveness, and weak upper airway motor control produce unstable breathing with periodic apnea, while immature hypothalamic and sympathetic pathways limit vasomotor tone and brown-fat thermogenesis, necessitating external thermal support. Immature central motor organization and reduced flexor tone also impair spontaneous midline posture and physiologic self-regulation.
The preterm brain simultaneously exhibits vascular and white matter vulnerability during this interval. Fragile germinal matrix vasculature predisposes to intraventricular hemorrhage, while pre-oligodendrocytes predominant between approximately 24–32 weeks of gestation are highly susceptible to hypoxia–ischemia and inflammation, creating a developmental window for periventricular leukomalacia. Fluctuating cerebral perfusion and oxygenation associated with respiratory and hemodynamic instability, therefore, affect both vascular integrity and white matter maturation.
These vulnerabilities define recommended neuroimaging and developmental care practices. Cranial ultrasonography during the first postnatal week and again at 7–14 days targets hemorrhage and early white matter injury. Developmental positioning— nesting, boundary support, and facilitated flexion—provides external containment that supports postural tone development and behavioral state regulation during immature central neuroregulatory function.
At approximately 33–35 weeks PMA, maturation of brainstem respiratory generators and autonomic pathways permits a stable respiratory rhythm, improved arousal responses, effective thermoregulation, and sustained flexed posture. Resolution of apnea and discontinuation of caffeine therapy around 34 weeks PMA reflect maturation of central respiratory control within this neuroregulatory window.
Kidney:
Nephrogenesis and tubular differentiation continue until approximately 34–36 weeks of gestation, rendering the preterm kidney structurally and functionally immature during early transition. Limited sodium reabsorption and relative chloride retention lower the plasma sodium–chloride difference, producing the mild hyperchloremic metabolic acidosis characteristic of prematurity. Immature filtration and autoregulation increase susceptibility to acute kidney injury during systemic instability.
These features define renal-directed monitoring and pharmacologic adjustment. Reduced glomerular filtration during the first postnatal week necessitates prolonged dosing intervals for renally cleared medications; intervals shorten as clearance matures. Electrolyte monitoring and sodium supplementation reflect maturation of tubular transport within this renal biologic window.
PHASE 2—INTERFACE:
Environmental Interaction and Barrier Maturation (~28–34 wk PMA):
Dominant systems: Skin, Gut–Immune, Retina:
As lung–cardiovascular stability improves, the infant’s physiologic interaction with the external environment expands. Organs mediating exchange with that environment—skin, intestine, immune system, and retina—become the principal sites of vulnerability. Care focus shifts toward the regulation of the environmental interface and barrier function.
Skin:
The epidermal barrier develops during the third trimester through keratinization and intercellular lipid deposition. Extremely preterm infants have minimal stratum corneum and high permeability, producing substantial insensible water and heat loss. Because environmental exposure begins immediately at birth while structural immaturity is maximal, cutaneous vulnerability peaks earliest among interface organs. Barrier formation accelerates between approximately 30–34 wk PMA, reducing permeability and stabilizing fluid and thermal balance.
Gut and Neurointestinal Maturation:
Intestinal epithelial integrity, mucosal immune regulation, enteric nervous system function, smooth-muscle motility, and neurogastrointestinal coordination mature progressively through approximately 28–34 weeks PMA. Preterm birth exposes the intestine to enteral nutrients and microbial colonization while neuromuscular coordination and barrier function remain incomplete, defining the intestinal biologic window.
Neurointestinal motility is most immature in extremely preterm infants (<28 weeks PMA) with weak peristalsis, delayed gastric emptying, and poor coordination of swallowing and breathing. During this stage, feeds are typically minimal and trophic. As feeding volumes advance between approximately 28 and 32 weeks PMA, while coordination remains incomplete, delayed gastric emptying and ineffective peristalsis become clinically evident as gastric residuals, abdominal distension, and feeding intolerance. Continued maturation after approximately 32–34 weeks PMA permits progressive improvement in gastric emptying and tolerance.
Intestinal epithelial and immune barrier maturation during approximately 28–34 weeks of gestation permits increased interaction with the luminal microbiota. Immature tight-junction integrity and dysregulated inflammatory responses predispose to necrotizing enterocolitis, which typically emerges after several weeks of feeding and colonization and therefore peaks within this biologic window.
Oral feeding readiness emerges as neurogastrointestinal and neurorespiratory coordination mature during late prematurity. Coordinated sucking, swallowing, and breathing develop primarily between approximately 32 and 34 weeks PMA, with progressive improvement after approximately 34–36 weeks PMA. Supportive feeding therapy practices optimize bolus control while coordination remains incomplete.
Retina:
Retinal vascularization progresses during 28–40 weeks of gestation under oxygen-regulated signaling. Premature hyperoxia suppresses physiologic angiogenesis early, leaving the peripheral retina avascular. As metabolic demand rises later (~30–34 wk PMA), hypoxia drives pathological neovascularization characteristic of retinopathy of prematurity, peaking at 34–38 wk PMA. An initial retinal examination at approximately 31 wk PMA (4weeks postnatal age) targets the onset of this proliferative phase.
PHASE 3—GROWTH ACCRETION:
Nutrient-dependent Construction (~30–36 wk PMA): Dominant systems: Bone–Mineral, Hematologic, Somatic Growth: With systemic stability largely achieved and environmental interfaces maturing, development becomes dominated by sustained tissue growth. Organs requiring high substrate supply— bone, hematopoietic system, and somatic tissues—now define vulnerability, and care priorities shift toward ensuring that nutrient supply matches growth demand.
Bone–Mineral:
Fetal skeletal mineralization accelerates after 28 weeks of gestation. Preterm birth interrupts placental mineral transfer while skeletal growth continues. As growth accelerates after approximately 30– 32 wk PMA, mineral requirement exceeds supply and metabolic bone disease emerges. Biochemical screening beginning around 4–6 weeks postnatal age corresponds to the onset of mineral accretion demand within this window.
Erythropoiesis transitions to marrow dominance during late gestation. After preterm birth, low erythropoietin production and rapid growth produce anemia of prematurity, with hemoglobin nadir near 32–34 wk PMA. Periodic hemoglobin monitoring during the first 6–8 weeks aligns with this erythroid accretion window.
Somatic Growth:
Rapid tissue accretion, normally occurring in late gestation, must occur postnatally in preterm infants. Immature anabolic regulation limits early growth; improving nutrition and endocrine maturation toward term PMA increases growth velocity.
PHASE 4 — NEAR-TERM NEUROINTEGRATIVE MATURATION:
Functional Synchronization (~34–40 wk PMA):
Dominant systems: Brainstem–Autonomic–GI Integration:
Near-term equivalent age: organ systems are largely structurally complete, and the dominant developmental task becomes integration. Brainstem respiratory rhythm, autonomic regulation, thermoregulation, postural stability, and neurogastrointestinal coordination converge toward mature function. Vulnerability now reflects incomplete coordination rather than structural immaturity.
Preterm infants approaching discharge may appear stable supine but may desaturate when positioned semi-upright. Car-seat tolerance testing evaluates this final stage of neurorespiratory integration. Gastroesophageal reflux and feeding discoordination likewise decline during this phase as coordination matures.
CLINICAL MEANING:
The biologic windows framework provides a developmental basis for anticipatory neonatal care. Because organ vulnerability follows maturation rather than chronological age alone, monitoring and management priorities should align with the dominant developmental processes at each PMA. Early transition care emphasizes stabilization of oxygenation, perfusion, and metabolic homeostasis to protect the immature lung, brain, and kidney. As cardiopulmonary stability improves, care shifts toward regulation of environmental interfaces, including thermal balance, feeding progression, microbial exposure, and oxygen stability. With advancing maturation, sustained growth and substrate delivery become central to preventing deficits in mineralization, erythropoiesis, and somatic accretion. Near-term equivalent age, assessment focuses on integrated physiologic competence, including respiratory control, feeding coordination, and autonomic stability before discharge.
Recognizing these maturation-linked priorities enables anticipatory monitoring, phase-appropriate interventions, and physiologically timed screening across the course of prematurity.
SUMMARY:
Biologic windows of prematurity arise because organ maturation proceeds in parallel during late gestation. Vulnerability shifts with PMA—from systemic stabilization (lung–brain–kidney), to environmental interface (skin–gut–retina), to growth-dependent accretion (bone–hematologic–somatic growth), and finally to neurointegrative maturation near term age. Interpreting prematurity through these sequential biologic windows provides a unified developmental framework linking maturation, disease timing, and neonatal management across the course of prematurity.

Abstract
Importance: Contradictory differences regarding fertility among adults born very preterm (VP; <32 weeks’ gestation) or with very low birth weight (VLBW; <1500 g) have been reported at different ages. Longitudinal investigations of fertility beyond ages 20 to 29 years are lacking.
Objective: To assess whether VP and VLBW are associated with lower fertility up to age 35 years.
Design, setting, and participants: This prospective population-based cohort study was conducted in Bavaria, Germany, with participants born between January 1985 and March 1986 who required admission to a children’s hospital within the first 10 days after birth. Healthy infants born at term (ie, at least 37 weeks’ gestation) in the same hospital and children alive at 6 years were selected as controls. Follow-up occurred repeatedly up to 34 to 35 years. Data were analyzed from July to December 2024.
Exposures: Gestational age and birth weight.
Main outcomes and measures: Cumulative incidence of having the first alive child (ie, fertility) was calculated using Kaplan-Meier estimation and the difference between participants born VP or with VLBW and term-born participants was compared with log-rank tests. Univariable and hierarchical multivariable Cox proportional hazards regressions were performed to estimate hazard ratios (HRs) for fertility associated with VP and VLBW adjusting for individual factors. Estimations were compared between early (<30 years) and late (≥30 years) reproductive windows.
Results: A total of 414 participants (212 born VP or with VLBW and 202 term-born) were included (mean [SD] age, 34.67 [0.53] years; 216 [52.2%] female). Cumulative incidence of having the first alive child was lower in participants born VP or with VLBW throughout the follow-up. The association of VP and VLBW with lower fertility was significant during the late (HR, 0.46; 95% CI, 0.31-0.68) but not early (HR, 0.73; 95% CI, 0.47-1.14) reproductive window; this association attenuated after adjusting for neonatal factors (sex and family socioeconomic status) and childhood neurosensory impairment at step 2 (HR, 0.59; 95% CI 0.40-0.88), childhood family factors (eg, parenting) at step 3 (HR, 0.61; 95% CI, 0.40-0.92), and there was no longer an association after adjusting for sociodemographic factors (eg, partnering) at step 4 (HR, 0.78; 95% CI, 0.50-1.20). Partnering (ie, being married or cohabitating with a partner) had the largest magnitude association with fertility across both the early (HR, 7.87; 95% CI, 3.44-18.00) and late (HR, 3.95; 95% CI, 2.47-6.31) reproductive windows.
Conclusions and relevance: In this cohort study of participants born VP or with VLBW and term-born adults, VP and VLBW were associated with overall lower fertility. A major reason was an association of less partnering with lower fertility, suggesting that partnering is key to reproductive success.
https://pubmed.ncbi.nlm.nih.gov/40105837/




https://www.frontiersin.org/journals/education/articles/10.3389/feduc.2026.1758593/full



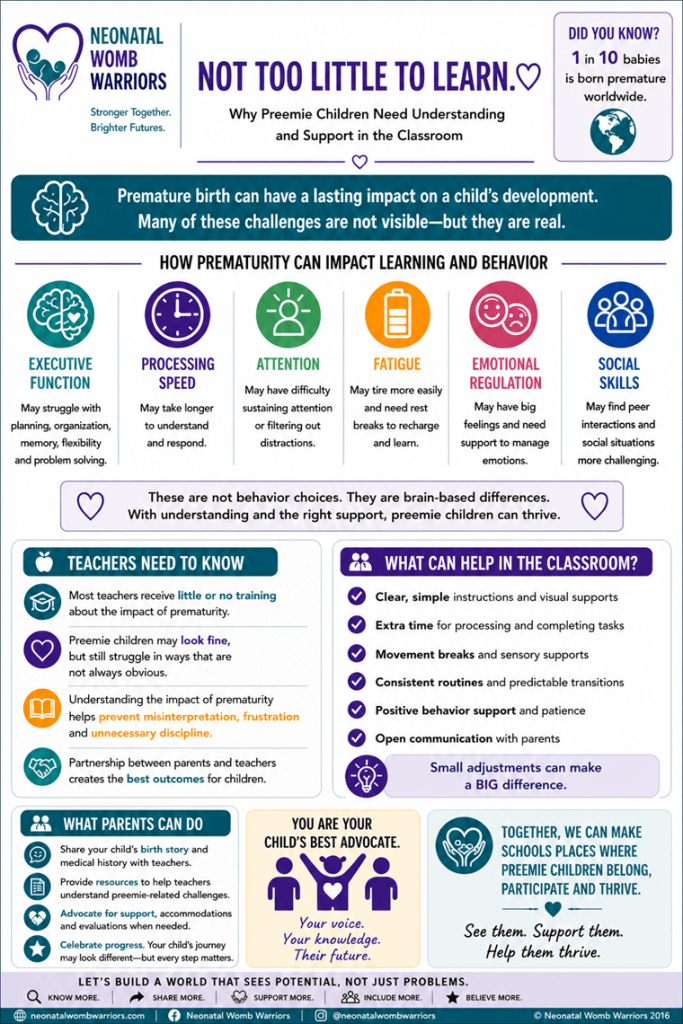
by Neonatal Womb Warriors (2026) using AI-assisted design tools (Informal infographic/educational resource). Content informed by global research on preterm birth and education. This infographic is intended for educational purposes and does not replace professional medical or educational advice.



https://pubmed.ncbi.nlm.nih.gov/40816932/

In 2021, Université de Montréal graduate student Adèle Saives asked to fast-track into the doctoral program in nursing specifically to work on an ambitious project: a device to support the development of prematurely newborn babies.
This was a major undertaking, probably a first for a doctorate in nursing, according to her supervisors, Faculty of Nursing professor Marjolaine Héon and Marilyn Aita, a professor and researcher at the Azrieli Research Centre of the UdeM-affiliated CHU Sainte-Justine.
Saives had come across a study of a handmade neonatal hammock used in Brazil. After some research, she concluded that nothing similar existed in North America or Europe, and there was no standardized model on the market.
She called her project Prémac — and the hammock has come a long way since. It underwent initial testing in the neonatal unit at CHU Sainte-Justine and then received funding under a pilot program there to clinically validate new healthcare technologies in a real-world environment.
The project is jointly supported by the CHU Sainte-Justine Research Centre, Quebec’s Ministry of Economy, Innovation and Energy, and Axelys, a non-profit that supports the transfer of innovations produced by research to promote sustainable prosperity for Quebec.
Developed by a research team led by Adèle Saives who is supported by a CIHR doctoral training scholarship, and used in the neonatal unit at CHU Sainte-Justine, the hammock is already generating significant interest. Since a patent application for it is still under review, however, images of it are not being circulated.
Reproducing intrauterine movement
After delivery, a premature baby is flung into a blindingly different environment.
“In neonatal units, we want to optimize neurological development,” said Saives, who has also worked as a neonatal nurse at CHU Sainte-Justine since 2020. “So we try to provide the same sensory stimulation as during pregnancy.”
In the womb, it’s darker and quieter than outside, but the foetus is also free to move. “That provides important stimuli for brain development, promoting everything from coordination to sleep,” Saives said.
In an incubator, however, premature babies have little opportunity to move in ways that engage their vestibular system. Saives wanted to create a hammock that would allow more movement, activated simply by the baby’s breathing, while at the same time enveloping the baby.
“This is the closest we can get to in utero movement,” she said. “We think it could have positive effects on the stability of physiological parameters and also on sleep, which is very important for the development of premature newborns.”
Input from many quarters
Saives drew extensively on her experience and practical knowledge as a nurse.
“Adèle did it all,” said Aita and Héon. “She didn’t just come up with the idea, she created the design using software, chose the materials, sewed the prototype and recruited the hospital’s seamstresses to produce the final models.”
Saives was also concerned with getting her idea accepted. “It was important to make my project compatible with the care routine in a clinical setting,” she said. The response at the hospital was enthusiastic from day one, she added.
Saives consulted people from a variety of disciplines: neonatologists, physiotherapists, occupational therapists, respiratory therapists. She then worked on the first sketches with Sophie Labat, then a doctoral student in biomedical engineering at Polytechnique Montréal.
Then she sewed the prototype. To design a fastener that would work on the new-model incubator used in the neonatal unit at CHU Sainte-Justine, she sought the expertise of Julie Lévesque, an engineer at the Centre de métallurgie du Québec.
While the project spans several disciplines, Saives’ experience gave her a holistic vision. “Her university training in nursing equipped her to consider many aspects of the device in terms of developmental care,” said Aita.
Every detail was carefully analyzed, from the choice of the material that gently envelops the babies to the seams, which must not damage their fragile skin. The hammock can also be quickly laid flat for an emergency intervention and can be easily disinfected.
The real test
After confirming the safety of the technology, Saives is now conducting a randomized clinical trial involving premature babies in the neonatal unit at CHU Sainte-Justine who were born at between 26 and 35 weeks.
“I want to evaluate the effects of the hammock compared with babies lying on their backs in an incubator,” she explained.
In addition to the funding agencies and Axelys, Saives has received assistance from other organizations including the Canadian Institutes of Health Research, UdeM’s Bureau Recherche-Développement-Valorisation (BRDV) and the CHU Sainte-Justine’s Bureau de l’innovation.
“This was a new process for us,” said Saives. “We really appreciated the step-by-step support.”
If the results of the clinical trial are positive, Saives hopes to patent the Prémac and see widespread use of the device. But whatever the results, the project has been formative and rich in both intellectual and human discoveries, she said.













