
MONTENEGRO

PRETERM BIRTH RATES – Montenegro
Rank: 109 –Rate: 9.2 Estimated # of preterm births per 100 live births
(USA – 12 %, Global Average: 11.1%)
Montenegro is a country in southeast Europe on the Adriatic coast of the Balkans. It borders Bosnia and Herzegovina to the north, Serbia to the east, Albania to the southeast, the Adriatic Sea and Croatia to the west. The largest and capital city Podgorica covers 10.4% of Montenegro’s territory and is home to 29.9% of the country’s population, while Cetinje has the status of old royal capital. Major group in the country are Montenegrins. Serbs form a significant minority at 28.7%, followed by Bosniaks, Albanians and Croats.
Classified by the World Bank as an upper middle-income country, Montenegro is a member of the UN, NATO, the World Trade Organization, the Organization for Security and Co-operation in Europe, the Council of Europe, and the Central European Free Trade Agreement. Montenegro is a founding member of the Union for the Mediterranean. It is also in the process of joining the European Union.
According to the Health Consumer Powerhouse the country has “the most backward health system in Europe”. Public services are financed through the Health Insurance Fund. It is funded by payroll contributions of 10.5%. About 5% of the national budget is allocated to healthcare. Only €5 million was provided for all public hospital supplies in 2016, about a third of what was thought necessary. 72.5% of total health spending comes from the fund. Most of the rest is direct out-of-pocket payments.
The Ministry of Health in Montenegro guides a national health fund. Contributions of employer’s and employee’s entitles citizens to health care. This program covers most medical services, except from particular physicians.[32]
Source: https://en.wikipedia.org/wiki/Montenegro

COMMUNITY
As we look forward to celebrating the beginning of our 6th year (February) of sharing this “blog” we thank all Neonatal Womb Warrior/Preterm Birth Community members for your presence, and for the courage, endurance, intelligence, creativity and compassion you share with All experiencing this challenging life journey. Knowing you are out there we feel a sacred kinship and energetic connection that only a soul-centered awareness can create. From deep within our hearts, with Joy and Gratitude, we (Kat and Kathy) Thank You.

Queen Elizabeth II honours Inga Warren
Posted on 22 January 2021


© privat
This is one of the highest accolades given and is a wonderful recognition, justly deserved, of her work and dedication to babies and their families.
Inga Warren is a neonatal development specialist with extensive experience as an occupational therapist working with children of all ages and families. She teaches nurses, doctors and therapists to understand babies behaviour, their cues and body language, helping them to change practice and to involve parents in the care of their child. She is also the director of the UK NIDCAP (Newborn Individualized Developmental Care and Assessment Program )Training Centre.
Together with Monique Oude Reimer-van Kilsdonk, Inga Warren developed the FINE (Family and Infant Neurodevelopmental Education) programme – an educational pathway in infant and family-centred developmental care for all healthcare professionals working in neonatal care. She has been a supporter of EFCNI’s work for many years and was also very much involved in the European Standards of Care for Newborn health project where she contributed with her knowledge to the standard topic of Infant and family centred developmental care.
Source: https://www.efcni.org/news/queen-elizabeth-ii-honours-inga-warren-from-nidcap/

A large study of UK healthcare workers finds that immunity after coronavirus infection lasts for months, but those with antibodies may still be able to carry and spread the virus upon re-exposure.

Max Kozlov Jan 14, 2021
Immune responses from a previous SARS-CoV-2 infection reduce survivors’ risk of reinfection by more than 83 percent for at least five months, according to preliminary data from a study of more than 20,000 UK healthcare workers published by Public Health England. The researchers caution that people previously infected may still be able to transmit the virus.
“Overall I think this is good news,” Imperial College London epidemiologist Susan Hopkins, a senior medical adviser to Public Health England (PHE), tells The Guardian. “It allows people to feel that prior infection will protect them from future infections, but at the same time it is not complete protection, and therefore they still need to be careful when they are out and about.”
Between June and November last year, the researchers monitored, through monthly serological tests and PCR tests twice a month, the infection rates in those who had been infected with the virus before June and those who had not. They found 44 potential reinfections, including 13 symptomatic cases, among the 6,614 who’d had the virus before, and 318 cases among the 14,173 who had no evidence of past infection. The authors concluded from these results that prior exposure to SARS-CoV-2 provides 94 percent protection against symptomatic reinfection, and 75 percent protection against asymptomatic reinfection.
The researchers also found that people who become reinfected can carry a high viral load in their noses and throats, even in asymptomatic cases, which correlates with a higher risk of spreading the virus to others, says Hopkins.
“Reinfection is pretty unusual, so that’s good news,” University of Pennsylvania immunologist John Wherry tells Nature. “But you’re not free to run around without a mask.”
“The immunity gives you a similar effect to the Pfizer vaccine and a much better effect than the AstraZeneca vaccine and that is reassuring for people. But we still see people who could transmit and so we want to strike a note of caution,” Hopkins tells The Guardian. In clinical trials, two doses of the Pfizer vaccine were 95 percent effective at preventing infection, compared with 62 percent from two doses of the Oxford/AstraZeneca vaccine. The Pfizer vaccine has been approved for emergency use in the UK, Canada, Mexico, the US, Switzerland, and the EU, while the Oxford/AstraZeneca vaccine has been approved for emergency use in the UK, Argentina, India, and Mexico.
None of the individuals with potential reinfections had PCR-based evidence of a first infection, but all harbored antibodies against SARS-CoV-2 at the outset of the study, which has not yet been peer-reviewed or published in any medical journal. The authors tried to measure antibodies that were specific to SARS-CoV-2, but prior studies show that antibodies against other coronaviruses may cross-react to show a false positive result for SARS-CoV-2 antibodies, which is why the researchers termed these cases “possible reinfections.”
The PHE team does not yet have enough data to understand who might be at highest risk of reinfection. Francis Crick Institute immunologist George Kassiotis tells Nature that participants in the study were mainly women, and mostly under the age of 60. “This group is unlikely to experience the most severe form of COVID-19,” he says, “and may not be representative of the population as a whole.”
Correction (January 15): We omitted Mexico from the list of countries that have approved the Pfizer vaccine. The Scientist regrets the error.

Mapping neonatal and under 5 mortality in India

Published : May 11, 2020 Praaveen Kumar, Nalini Singhal
India is one of the world’s largest and most populous countries, made up of more than 700 diverse districts. Variations in mortality in the country are known at the macro level, and now the India State-Level Disease Burden Initiative Child Mortality Collaborators have mapped neonatal and under-5 mortality rates from 2000 to 2017 for every district in India, going down to geospatial grids as small as 5 km × 5 km. In The Lancet, the study authors report that the under-5 mortality rate (U5MR) in India decreased from 83·1 deaths (95% uncertainty interval 76·7–90·1) in 2000 to 42·4 deaths (36·5–50·0) per 1000 livebirths in 2017, and the neonatal mortality rate (NMR) decreased from 38·0 deaths (34·2–41·6) to 23·5 deaths (20·1–27·8) per 1000 livebirths. U5MR varied 5·7 times between the various states and 10·5 times between the 723 districts in 2017, whereas NMR varied 4·5 times across the states and 8·0 times across the districts. Child and maternal malnutrition was the main risk factor, contributing to 68·2% (65·8–70·7) of under-5 deaths and 83·0% (80·6–85·0) of neonatal deaths in India in 2017, while 10·8% (9·1–12·4) of under-5 deaths could be attributed to unsafe water and sanitation and 8·8% (7·0–10·3) to air pollution. The authors found substantial variations between the states in the magnitude and rate of decline in neonatal and under-5 mortality, with higher variations between the districts. Additionally, inequality between districts has increased within the majority of the states.
The underlying system-based causes of neonatal and under-5 mortality need to be better understood. One size cannot fit all, especially in such a large country. Contextual microplanning is required at the lowest administrative unit level, which should be based on real data of deaths and their causes, available resources, and coverage and quality of services. The Article describes important differences in local outcomes that can inform the designing of strategies with local stakeholders in areas such as education, provision of equipment (eg, incubators, neonatal radiant warmers, neonatal resuscitation bag and masks, autoclaves), and transportation of mothers and neonates to health facilities to accelerate the decrease in NMR and U5MR. Local health-care providers understand the challenges their areas face and thus might be able to provide solutions for sustainable improvements. A district-level perinatal–neonatal care model driven by local coordination committees composed of all stakeholders has been proposed.
Quality care, knowledge translation to caregivers, and local implementation for delivery of health care are all key to survival of children and mothers.
Focusing on local changes using local data can lead to improved outcomes, as has been shown in Canada.
Health is a state responsibility; however, in India planning typically occurs at the central level with very little involvement of lower level providers. This study provides valuable information to highlight the importance of standardised national perinatal–neonatal surveillance data that can be turned into actionable information for end users at the lowest administrative unit.
The authors have brought attention to the fact that, with current trends, India is unlikely to achieve the Sustainable Development Goal (SDG) 2030 NMR targets or either of the National Health Policy 2025 targets.
To reach the SDG 2030 targets individually, 246 (34%) districts for U5MR and 430 (59%) districts for NMR would need a higher rate of improvement than they had up to 2017.
Simply knowing the causes is not enough. Structural deficiencies remain, with glaring bottlenecks. To provide adequate care, India needs 20 000–30 000 level 3 neonatal intensive care unit (NICU) beds and 75 000–100 000 level 2 special newborn care beds.
However, the numbers of beds available, especially level 3 NICU beds, are grossly inadequate.
It remains common in government hospitals to have up to five sick newborn babies sharing a cot.
In such scenarios, quality improvement efforts that target process improvement alone do not achieve their full potential benefits. The Indian Government’s flagship insurance scheme—the Ayushman Bharat Pradhan Mantri Jan Arogya Yojana—holds great promise of rapidly expanding the pool of hospital beds by enrolling existing private health-care facilities. However, the effect on availability of level 3 NICU beds is yet to be seen. Upgrading and operationalising the underused newborn stabilisation unit beds across the country along with a functional back-referral system could substantially augment the number of level 2 beds available.
The variation in district-wise mortality and its relationship to social development has caught the attention of planners. The Aspirational Districts Programme launched in 2018 focuses on the 115 lowest-performing districts for all-round development through convergence, collaboration, and competition. Intersectoral and centre-state convergence addresses education, skills development, financial inclusion, water, air, infrastructure, and other determinants of health. Apart from state-wise and district-wise differences, another source of huge variation is urban slums. The mortality indicators in urban slums are even worse than in rural areas and they have their own unique set of problems.
The growing inequalities shown in this study between 2000 and 2017, against a global aim of equity, are of concern and reflect the widening gap between the rich and the poor.
Finally, the estimates of the causes of neonatal deaths in the country need to be more robust for good planning. The study authors point out the limitations of verbal autopsy methods.
However, the medical certification of cause of death is not always credible, given the expertise of medical officers in the districts with limited laboratory support.
The authors make a strong case for local implementation and provide guidance to address gaps. In this direction, the Indian Government has launched programmes to improve nutritional outcomes for children, pregnant women, and lactating mothers; clean water and sanitation; and hygiene in public health facilities. With local input and planning, both U5MR and NMR can be brought down.
Source: https://www.thelancet.com/journals/lancet/article/PIIS0140-6736(20)31050-3/fulltext?rss=yes

Thunderstorm in Montenegro| Soothing Thunder & Rain Sounds For Sleep| Relaxation| Studying| 10 Hours
Jan 6, 2021 Outdoor Therapy
Relax, Study or FALL ASLEEP FAST with the soothing sounds of this thunderstorm in Montenegro.
PREEMIE FAMILY PARTNERS

Beating the odds: The world’s most premature twins to celebrate 2nd birthday
Sarah Kay LeBlanc Des Moines Register
DES MOINES, Iowa — Shorter than a Barbie doll and lighter than a football, Kambry Ewoldt entered the world fighting to survive.
Kambry and her identical twin sister, Keeley, were born Nov. 24, 2018, around the 22-week mark of the pregnancy of their mother, Jade Ewoldt. They weighed 15.8 ounces and 1 pound 1.3 ounces, respectively, and spent the first four months of their lives in the Neonatal Intensive Care Unit at the University of Iowa Stead Family Children’s Hospital before they could go home.
Guinness World Records has recognized them as the world’s most premature twins.
Today, the girls love singing “Baby Shark,” doing the Chicken Dance and painting pictures. They have their own personalities — Kambry is more of a tomboy and Keeley is very girly, Ewoldt said — and are excited to celebrate their second birthdays.
It’s a milestone they weren’t guaranteed.
‘Survival mode’
Ewoldt, already a mother of two, knew having twins meant it would be a high-risk pregnancy.
At 16 weeks, doctors told Ewoldt her daughters had twin-to-twin transfusion syndrome, where they were sharing blood through blood vessels in the womb. If untreated, the syndrome can be deadly to babies.
″(TTTS) is also very rare,” Dr. Jonathan Klein, a neonatologist and medical director of the NICU at University of Iowa Stead Family Children’s Hospital, told The Associated Press in January 2019, about two months after the Ewoldt twins were born. “A lot of patients pass away before they are even born.”
At 17 weeks, Ewoldt underwent surgery at Cincinnati Children’s Hospital to seal and disconnect the twins’ aberrant vessels.
Most mothers deliver their babies within 10 weeks of the surgery, and Ewoldt was no exception — Keeley and Kambry were born about a month later on Nov. 24.
For the next five months, Ewoldt was split between two worlds: the hospital and home.
She made a two-hour round-trip commute almost every day from her home in Dysart — and her two older children, Koy and Kollins — to her newborn daughters. Constantly driving back and forth, she said, put her in “survival mode.”
“It was hard to leave the NICU knowing that I was having to compartmentalize life,” she said. “Leaving behind the twins, knowing I couldn’t take them home was painful and then (I was) going home to be with my other kids and shutting off thinking about the twins when I was with them.”
Stay safe and informed with updates on the spread of the coronavirus
As tiny infants, Kambry and Keeley were diagnosed with severe bronchopulmonary dysplasia, a chronic lung disease that makes breathing difficult. They have had to receive oxygen through nasal cannulas almost their entire lives, but were able to be taken off oxygen earlier this month.
Klein told the Des Moines Register, part of the USA TODAY Network, that even though the babies missed most of their lung development in utero by being born so early, they have done “extremely well” in their development.
“I would consider anytime babies like this on the cusp of viability survive, that it’s a pretty amazing situation, and it’s a huge dedication to a large team,” he said.
Life didn’t stop throwing obstacles in the Ewoldts’ path when Jade was able to bring her babies home. The twins are more susceptible to illness and last year had six hospital stays for the common cold.
“Something you or I would get the sniffles over would put them in the ICU,” Ewoldt said.
Although COVID-19 poses a significant risk to the twins, she said the family was already taking precautions against any sickness by staying indoors most of the time. It’s not perfect, but at least her family is together.
“I still feel torn between the two sets of kids, but at least I know, at the end of the day, the older kids get to do normal things where the twins get to stay healthy and I don’t have to decide between the two,” she said.
Giving thanks
November is a month full of meaning for the family. It’s the twins’ birthday month, and the birthday month for their older sister, Kollins, who will turn 5 on Nov. 30. World Prematurity Day, a day created by March of Dimes to support families of premature babies, was Nov. 17, and November is Prematurity Awareness Month.
It’s also the month Ewoldt said goodbye to her sister Baylee Hess, who died in a crash last year on Kollins’ birthday as she was driving to her parents’ house to watch movies with her mom. Hess, 26, collided with a tractor-trailer, and died at the scene.
As the family celebrates the twins at home this year, their birthday will be dedicated to Baylee, Ewoldt said.
“This is a month of many emotions, but I want to practice Thanksgiving,” she wrote in a Nov. 1 Facebook post on her page called “Keeley and Kambry’s Tribe.” “I’m thankful for the uneven road that brought us here even when I do not understand.”
With nearly 10,000 Facebook followers, Ewoldt hopes their story can reach and support families going through similar struggles.
Years before she was pregnant with twins, without knowing the information may help save her future children, Ewoldt saw a story about a family that did not have the opportunity to intervene when they received a twin-to-twin transfusion syndrome diagnosis and lost their children. When she received the same diagnosis, that story affected her decisions, she said. She hopes to pay it forward by sharing the knowledge she has gained.
“If our story can help save another baby, then it’s really important to continue to share,” she said.
Contributing: Associated Press
Source: World’s most premature twins to turn 2 in Iowa: Guinness World Records (usatoday.com)

For premature babies

Dr. Nils Bergman, Jill Bergman
Kangaroo Mother Care (KMC) has often been used as a treatment for premature or preterm babies.
In a rural third world situation where no incubators are available this method of caring for preterm or low birth weight babies can be life-saving. This works because the baby on the mother’s chest does not get cold, so the lungs function better. The baby in skin-to-skin contact also feels safe with mum’s familiar heart beat and voice so they do not become stressed and the heart rate, blood pressure and breathing stabilize faster. On the mother’s chest the baby also stimulates the production of breastmilk. This milk is vital for providing the preterm baby with the exact food needed to grow her brain.
About 4 million babies die each year in the first week of life, KMC could help 25% survive.
In a first world hospital setting KMC is also being used for premature babies. The same biology applies and means that the baby is more stable with all of the above benefits.
Any needed or available technology can be added when the baby is in skin to skin contact on mum’s chest.
For both contexts it is not just SURVIVAL of the preterm baby, but it is also the QUALITY of that survival in terms of brain growth, healthy brain wiring, emotional connectedness, bonding and attachment. KMC helps the physical, emotional, and social development to be the best it possibly can be!
When to do skin-to-skin contact:
- Skin-to-skin contact should be for every baby at birth.
- It is even more important for premature babies to help them stabilise.
- You can do skin-to-skin contact when you are sleeping, or walking around.
- You can do skin-to-skin contact with a baby on a ventilator or other machines
Some countries use Kangaroo Care only as an add-on to incubator care as a bit of cuddling for 10-60 minutes a day which helps mum and baby to bond together. This does help the mother produce more breast milk ! But if you hold your baby for less than an hour or full sleep awake cycle, it gives no benefit to the baby and might even do harm. Some places only allow you to hold your baby once she is “stable” and a particular weight and off all monitors and machines……………………advice???
Some say that the baby must stabilise in the incubator first and then have skin-to-skin contact. But this is a problem as babies do not stabilise in an incubator for days, they do stabilise in skin-to-skin contact.
Skin-to-skin contact should ideally be 24 hours a day so that your baby is never separated from you, her Mum. In this way stress is minimized. Some hospitals do not have enough space for mum’s to sleep in the hospital, but now that you know the importance of her brain wiring properly you will make it a priority to spend as much time as possible. The ideal is for mum and dad to take it in turns for the full 24 hours!
So take leave or get home help or whatever is needed to give your prem your support – she is fighting for her life and she needs you to help her. You will never regret this time that you spend with her.
The smaller the prem, the more they need their mother’s chest to stabilise even if they need medical technology as well.
KMC for Premature and Low Birth Weight Babies
The care of premature and low birth weight infants must be under the supervision of a qualified health professional if at all possible.
The KangaCarrier was originally designed to enable mothers of premature babies to be permanent incubators for their babies. Medical research has shown that maternal infant skin-to-skin contact is better than incubators for keeping babies warm, provides better breathing and heart rate, better breastfeeding and better growth. The problem is a mother has a preconceived idea of an incubator as something stuck in a hospital ward and not allowed to be moved or to be touched. The mother has been an incubator for nine months, and with the KangaCarrier she can now carry on being an incubator. Many premature and low birth weight babies will have special needs and require medical care, all these can be provided together with continuous skin-to-skin contact.
Skin-to-skin at birth
Immediately after birth, the low birth weight baby should be dried, placed on mother’s abdomen or chest, and covered with a cloth or blanket, just as described for a full term baby. Routine midwifery care and medical assessment should be completed speedily, with the newborn remaining on mother. Observations and monitoring will determine what medical support should be provided, and how this should be done.
This may in our current contexts require that the baby be separated, which must be accepted. A very premature infant may be too physically immature to exhibit the self-attachment behaviours of the full term baby, but the opportunity for early skin-to-skin contact is important nevertheless.
A premature baby will need help to breastfeed. Colostrum should be expressed and given by spoon or cup, or by gavage (tube) of necessary. For premature newborns, mothers should recline at an angle of 30 degrees, and should avoid moving about too much. This helps the baby’s breathing and sense of balance. Newborns should be stimulated as little as possible in this period.
After the first hour
Many premature babies will be stable in skin-to-skin contact after 90 minutes, often to the surprise of health professionals. Decisions need to be made about feeding and fluids and continued care. Oxygen by mask or by CPAP can easily be arranged while in skin-to-skin contact, drips and feeding tubes can be secured sideways.
If the baby is stable and the mother well, the KangaCarrier can be used just as described for full term babies. An important difference is that a premature baby should not be fed on demand, it needs to be fed far more frequently, every hour or two, day and night. This means loosening the wrapper regularly, so that mother can express breast milk. This is good for the baby and mother, though baby must never get cold.
The first six weeks
Many premature babies and low birth weight babies need intensive medical care, and may require care in incubators (6). Most neonatal intensive care units now allow mothers to start KMC for an hour or two a day, and build up the time successively as the baby gets better and the mother more confident. Even an hour a day has positive effects on baby, and just ten minutes a day can increase mother’s milk supply!
Once the baby is stable and gaining weight the KangaCarrier can be used day and night, both sleeping and walking. This is what it was originally designed for!
After six weeks
“How long should the premature baby stay this way?”
Again, no two babies are the same, and in this case the answer depends on a number of factors. But until the baby weighs 1800g, providing an ongoing heat source is physiologically essential, and even up to 2.2 kg a baby will easily become hypothermic. Beyond that weight, consider the baby as full term and apply the advice as above!
Once a baby reaches 4 kg or thereabouts, the KangaCarrier can be replaced by a sling or by the traditional African method of carrying on the back.
The benefits of KMC for premature babies
Skin-to-skin contact benefits for babies:
- Better brain development
- Better emotional development.
- Less stress
- Less crying
- Less brain bleeds
- More settled sleep
- Babies are more alert when they are awake
- babies feel less pain from injections
- The heart rate stabilises
- Oxygen saturation is more stable
- Less apnea attacks
- Breathe better
- The temperature is most stable on the mother
- Breastfeeding starts more easily
- More breastmilk is produced
- Gestation specific milk is produced
- Faster weight gain
- Baby can usually go home earlier
Skin-to-skin contact benefits for parents:
- Parents become central to the caring team
- Better bonding and interact with their child better
- Emotional healing
- Less guilt
- Parents are calmer
- Mum and dad are empowered and more confident
- Parents are able to learn their baby’s unique cues for hunger
- Parents and baby get more sleep
- Parents, (especially mothers)are less depressed
- Cope better in NICU
- See baby as less “abnormal”
Source: https://kangaroomothercare.com/about-kmc/for-premature-babies/
When a Baby’s Head is Misshapen: Positional Skull Deformities
When a baby spends a lot of time in one position, it can cause the shape of their head to change. This is called a positional skull deformity.
For about 20% of babies, a positional skull deformity occurs when they are in the womb or in the birth canal. More often, it happens in the first 4 to 12 weeks of life. This is when babies are not able to sit up or move on their own. By 6 months of age, many babies become more mobile and can turn their heads on their own more regularly.
What causes a baby’s head shape to change?
A baby’s skull has soft, bony plates that haven’t yet fused together. The bony plates can move a bit, which helps the baby’s head to pass through the birth canal. The plates also allow room for the brain to grow during the first year of life.
There are many things that can cause a positional skull deformity, such as:
- Preferred head position. Some babies like sitting or sleeping with their heads turned a certain way.
- Not enough tummy time. Tummy time is for babies who are awake and being watched. It helps babies master basic milestones like head lifting, turning over, sitting up, and crawling.
- Twin or triplet. Cramped or unusual positions in the womb can lead to changes in head shape.
- Prematurity. Lying flat on their backs in the hospital can cause a preemie’s head to become misshapen. Preemies also have softer bones that are not as fully formed as the bones of full-term babies.
- Birth complications. The baby’s position when moving through the birth canal can affect his or her head shape. Some other things that affect a baby’s head shape at birth are using forceps, vacuum extraction, or even a difficult labor.
- Torticollis. An estimated 85% of babies with torticollis, a condition where the neck muscles are tight or imbalanced, have a positional skull deformity. Infants who have torticollis also will need physical therapy.
3 common misshapen head types
A baby’s head shape change usually is related to the position in which they spend the most time. Your pediatrician can determine whether your baby’s head shape is caused by a positional skull deformity or an uncommon but more serious condition called craniosynostosis. Common positional skull deformities do not require surgery.
- Deformational brachycephaly is when the head is symmetrically (evenly) flat in the back and wide from side to side. The bone above the ears might seem to stick out. This is often seen in babies who spend a lot of time on their backs and do not get enough tummy time.
- Deformational plagiocephaly is when the head is asymmetrically (unevenly) flat in the back on one side. The forehead may be more prominent causing the head to look like a parallelogram. The ear may also shift forward on the flat side. This is often seen in babies who prefer to sleep with their heads turned to one side and those with torticollis.
- NICUcephaly is a common condition in preemies who spend their first few months of life in the neonatal intensive care unit (NICU). This positional skull deformity causes preemies to develop long, narrow heads.
A good time to check your baby’s head is after bath time when his or her hair is wet.
- The back of your baby’s head should be evenly round.
- Your baby’s ears should be even.
- The width of your baby’s head and forehead should be even and balanced.
If you notice any changes or have any concerns, talk to your pediatrician.
What if my baby has a positional skull deformity?
The best treatment is to prevent a positional skull deformity. And when it is found early, simple changes to your baby’s position will help. For example:
- Avoid too much time in a car seat, bouncy seat, baby swing, or other carrier. These positions put pressure on the back of your baby’s head.
- Increase tummy time. It is important to put babies on their backs to sleep. But a baby needs supervised time on their tummy to play during the day. This also lets your baby exercise their neck, back, shoulders, arms and hips. Start with short spurts of time. Gradually work up to an hour per day in several short sessions.
- Switch or alternate arms when holding and feeding your baby.
- Alternate which end you of the crib you place your baby down for sleep.
Positional skull deformities do not affect brain growth or intellectual development. They are purely cosmetic, and the majority do not require surgery.
Early intervention & therapy
In some cases, your baby’s doctor may recommend treatment for positional skull deformity, particularly those with moderate or severe flattening.
Physical therapy. Your pediatrician may refer your baby for early intervention services and an evaluation from a pediatric physical therapist. The therapist will check your baby for delayed motor skills caused by poor head and neck control, and for torticollis. You will also learn stretching and positioning exercises to do with your baby at home. Depending on how severe the positional skull deformity is, your baby may need weekly therapy.
Helmet therapy. If your baby has moderate or severe flattening that does not respond to treatment by 5 or 6 months of age, he or she may need helmet therapy. Molding helmets work by re-shaping the baby’s head and are fitted by a specialist.
Surgery should only be considered when all other options have been exhausted, and after consulting with a neurosurgeon or pediatric plastic surgeon who specializes in these types of cases.
Remember: Talk with your pediatrician if you have any questions or concerns about your baby’s growth and development.
Last Updated 8/31/2020 Source -American Academy of Pediatrics Section on Neurological Surgery and Section on Plastic and Reconstructive Surgery (Copyright © 2020)

HEALTHCARE PARTNERS
NICU Collaboration During the COVID-19 Worldwide Pandemic: A Memory of Gratitude
Marana, Joseph MEd, MSN, RNC-NIC; Manager
Advances in Neonatal Care: December 2020 – Volume 20 – Issue 6 – p 424-425
“Felicidades!” I exclaimed! And right there, I saw it.
Through the muffled voice of wearing two masks, a distant image through the face shield I was wearing, I saw the look to his daughter. It was nearly imperceptible. It was the slightest hesitation. To stare, and see, his infant for the very first time. In the rush of moment, installing the car seat base in the heat of the afternoon, with his mother-in-law, his infant’s nurse, and myself holding the infant in the car seat, along with a few bags of supplies for the infant, he only took a fraction of a second to do it. But right there, in that moment, it felt like time stopped. The air felt heavy, the sounds drowned out to silence, and as I father myself I recognized in his face what this was. It was a look of love.
I have seen it many times before. The first time a father lays his eyes on his newborn infant is always different. It’s always new. Usually, it is during or immediately after delivery. It is often without speech. But even if a picture is worth a thousand words, the image every time is indescribable. It is love.
Typically, the birth of an infant, especially in an operating room (OR), is filled with lots of people. Nurses taking care of the mother and nurses taking care of the infant. Nurse practitioners right there to “catch” the infant and wrap in a blanket, obstetricians and their team at the surgical site, and anesthesiologists at the head of the bed. The father anxiously standing by, peering on toes next to neonatologists at the ready. In this particular case, with this specific infant, instead of the father being there, it was me. It was us.
She was the first infant born in our COVID OR, in the specifically built COVID unit. The OR had been equipped and prepared for weeks, if not months, but doctors and nurses, practitioners, and respiratory therapists to develop plans for deliveries just like this one was. We had spent days setting up machines, placing beds, and staging trays and lights. And then we spent days and weeks rearranging and adjusting it all again, as we talked through situations and scenarios. For days, we had mapped out who of our staff was going to be working, who was going to be going to the delivery, who was going to do what, who was going to be where. It was my plan to switch places with the nurse who was supposed to be going, to minimize her risk of exposure and, hopefully, her anxiety. But as the confident and dutiful nurse she has always been, she refused to do anything less than to be right there for the infant. So by the time the COVID-positive mother was brought in, they closed the doors and it was us two. Next to our neonatologist, the 3 of us were the neonatal intensive care unit (NICU) team ready to receive and, if necessary, stabilize and resuscitate this little baby girl.
The room was a flurry of action. With assured confidence and precision, I admired the Labor & Delivery staff who worked quickly and efficiently to make sure everything was set. It was as if they had done this thousands of times before. The thing was they had. Just never like this. In a negative pressure OR, with noisy HEPA filters, a smaller than usual space, and personal protective equipment (PPE) covering our mouths and noses, our eyes, our faces, our hair, bodies, and even shoes, all of us were uniformly protected from any chance of exposure. I remember looking at the mother’s face. Calm, eyes closed.
When the infant was born, instead of her father present, it was the physician and myself, alongside her nurse, the first to see her, the first to touch her, the first to talk to her. It was decided that she would be admitted to the NICU. So as I assisted in unplugging and unlocking the enclosed isolette to transport, I couldn’t help but recognize that unlike most deliveries, the mother would not get the chance to hold her infant right away. As we exited the room, despite all the other things going on, the Labor & Delivery staff moved poles and carts to clear a path. As we came out of the room, there were more of our team members outside, ready to assist with whatever they could. Our NICU charge nurse took the lead, as we took off and put on more PPE since we were leaving the room. Like this infant’s own personal entourage, we walked to the exit of the unit where more NICU nurses and Respiratory Therapy were there to receive the bed, attach our support “shuttle” for additional oxygen and power support, and bring the whole production down to the NICU.
The next few days were new to us. The entire multidisciplinary team worked seamlessly to make sure this mother and family were updated. One of our nurses created a new Skype account for the mother to be able to video call and see her infant using her cell phone. Our nurse practitioners and provider team kept in constant contact to communicate plans for discharge. Social work was involved, all of our managers stayed in touch, and we used a newly purchased car seat to test the infant in, and then give to the family so they would not need to bring one in from home. Our medical director was involved, our neonatal nurse practitioner administrator helped facilitate any needs, and Nursing had the constant resources from our leadership in Maternal Child Health, our director, and even our chief nursing officer.
When the day of discharge came, the whole team worked to coordinate times for pickup, who would be coming into the hospital to meet us, where they would park, where the father would wait. The infant’s nurse that day spent the entire shift preparing enough bottles, diapers, discharge information, and even clothing to be as ready to go out into the world as any newborn infant could be. We put on our PPE and together, she and I took the infant down to the emergency department (ED). I coordinated with the manager of the ED, who made sure we had a place to go, and what the best way to go would be, as well as the best route for us and the family. In the humid heat of the afternoon, I stood at the entrance alongside our COVID screeners, ready to meet the father and escort the grandmother in.
I knew it was them when they showed up, the anxious father and his excited mother-in-law. “Hola abuela!” I said, “Me llamo Joe, soy enfermero con to nueva nina!” (Hi grandma! I’m Joe, a nurse with your new baby girl!). As the father waited outside, the nurse and I walked the grandmother through discharge paperwork, and finally gave her the infant to see, hold and now feed, for the very first time. ED nurses and even physicians stopped in their tracks as they walked by, smiling at the beautiful infant in her grandmother’s arms. After just a few minutes, it was time to walk out. Back through the corridors, we all exited into the heat. Again, this little infant’s own personal entourage. I gave the father the base of the car seat and I could see him struggling, and sweating to get it secured in.
And as he turned, I extended the tiny infant in her car seat toward his waiting arms. “Felicidades!” I exclaimed. And right there, I saw it. In the heat of the outside, rather than the cool of the OR, into his hands instead of ours, this man recognized, and for the briefest of moments, stared at his new daughter for the first time. It doesn’t happen often, even for an NICU nurse to be there for both the delivery and also the discharge home, but this time it was me. In the moment that time stopped. In the moment of recognition, as a father myself in the eyes of another father, the look I have seen many times before, but new and amazing each time. It was a look of love.
For all of our planning, our weeks and days and sleepless nights. For our staff who worried but were always there. Through the stress of the world, for our team who demonstrated kindness and deep compassion. Through the endless meetings, the constant e-mails, through the questions and concerns and even through the tears, it was and always is moments like this that remind us of who we are.
Joseph Marana, MEd, MSN, RNC-NIC Manager Neonatal Intensive Care Unit & Pediatrics Mercy Medical Center Baltimore, Maryland
© 2020 The Author. Published by Wolters Kluwer Health, Inc. on behalf of the National Association of Neonatal Nurses
Source:https://journals.lww.com/advancesinneonatalcare/Fulltext/2020/12000/NICU_Collaboration_During_the_COVID_19_Worldwide.2.aspx
Psychological resilience during the COVID-19 lockdown
William D.S. Killgore,⁎ Emily C. Taylor, Sara A. Cloonan, and Natalie S. Dailey
Author information Article notes Copyright and License information Disclaimer
Psychiatry Res. 2020 Sep; 291: 113216.

Abstract
Some individuals are more psychologically resilient to adversity than others, an issue of great importance during the emerging mental health issues associated with the COVID-19 pandemic. To identify factors that may contribute to greater psychological resilience during the first weeks of the nation-wide lockdown efforts, we asked 1,004 U.S. adults to complete assessments of resilience, mental health, and daily behaviors and relationships. Average resilience was lower than published norms, but was greater among those who tended to get outside more often, exercise more, perceive more social support from family, friends, and significant others, sleep better, and pray more often. Psychological resilience in the face of the pandemic is related to modifiable factors.
Source: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7280133/

Neonatal data tech and video streaming help clinicians enhance care

The two technologies are linked with biomedical equipment, the EHR and the laboratory information system to help improve outcomes. By Bill Siwicki June 19, 2020
THE PROBLEM
“On average, a neonate undergoes 768 manipulations and 1,341 procedures during their hospital stay,” said Dr. Gautam Yadav, a pediatric physician at Kalawati Hospital in Rewari, in the Indian state of Haryana. “It is important to study the effect of these manipulations on the clinical outcome. We needed access to technology to noninvasively monitor critical patients in the NICU to ensure any early insights into patient condition.”
PROPOSAL
Kalawati Hospital sought out some unique technology from Child Health Imprints, a vendor of neonatal health data technology, along with live-streaming video technology from vendor Wowza.
The Child Health Imprints system includes a NEO device that fetches live physiological data from medical devices, and that combines with the Wowza live video-streaming technology that takes the data of the neonate through an attached camera. This data is made available to clinicians through a web platform, iNICU, for informed decision-making.

“With our technologies, video streaming of neonates is being annotated in real time with any changes in physiological or clinical state of patients.” Dr. Gautam Yadav, Kalawati Hospital
“The third layer, an analytics layer, has the capability of autonomously tagging the manipulations – touch-points to the neonate – in live videos, correlating the physiological signals and identifying the pattern of variability in physiological signal while the neonate is being manipulated,” Yadav explained. “Further, it also identifies the duration and frequency of these manipulations. The complete technology solution studies the impact of manipulations on neonatal care and its association with adverse outcomes.”
MEETING THE CHALLENGE
Kalawati Hospital has used the technology at the bedside of critical patients in the NICU. It is used by doctors and nurses for quality control and training.
“This technology is linked with data of various biomedical equipment in the NICU from GE, Philips, Draeger and Nihon Kohden,” Yadav explained. “It also is linked with the EHR and lab information system. The analysis layer of video streaming allows clinicians to see changes in physiological data – such as heart rate, respiratory rate and oxygen saturation – during different manipulations.”
RESULTS
Yadav offers four success-metric areas of using the combined neonatal data and video-streaming technologies that Kalawati Hospital is studying.
“It is hypothesized that in emerging countries, especially India, an oversight on nursing staff through an automated computer vision platform will have significant improvement in clinical outcomes,” he said. “We are still collecting data with live streaming and [an] integrated platform, and it will take us a few more months to publish improvement in clinical outcomes – on a statistically significant population – and get it peer reviewed.”
On another front is the movement index – tone and body postures. It is well documented that early symptoms of disease can be picked by monitoring body tone and movement indexes, he noted. Currently, this is done manually by doctors during daily rounds in two to three minutes. With the technologies, it is hoped that, with data across many NICU areas of both healthy and diseased patients, it will allow the platform to aid in early disease-identification in an autonomous manner, he said.
“Then there is the command center for remote management/telemedicine,” Yadav said. “Most of the telemedicine solutions do not have [the] synchronized temporal data of a patient’s physiology, video and its correlation with patient well-being. With our technologies, video streaming of neonates is being annotated in real time with any changes in [the] physiological or clinical state of patients. This will allow artificial intelligence and deep learning applications to further improve clinical outcomes.”
And finally, manipulation frequency and duration.
“We have been able to see the number and duration of manipulations – both invasive and noninvasive – on the neonates,” Yadav explained. “We have submitted the results in a peer-reviewed publication for sharing the same with other NICUs. This has allowed us to baseline data of our existing outcomes and enabled us to initiate quality improvement. This has also allowed us to build a staff education program for team members with non-planned care manipulations.”
ADVICE FOR OTHERS
“Video streaming in critical care can allow direct visualization to clinicians and help us in informed decision-making, including remote access,” Yadav said. “This has aided in better accuracy of manipulations and its relationship with vital sign data. This tool has allowed our clinical staff to determine their operational performance. It will potentially allow the NICU staff to determine if certain manipulations are associated with adverse outcomes.”
Dr. Nils Bergman – KMC: Physiological response, cultural & practical challenges in field settings
Läkare Utan Gränser Nov 14, 2016

INNOVATIONS

Infographic: What Social Isolation Can Mean for the Brain

People who show low social engagement over long periods of time often show reductions in cognitive function. Studies of the brain may provide clues about this correlation.

Catherine Offord – Jul 13, 2020
Studies of animals and people experiencing isolation have identified several brain structures that appear to be affected by a lack of social interaction. Although these studies can’t identify causal relationships—and don’t always agree with one another—they shine a light on some of the mechanisms by which physical isolation, or feelings of loneliness, could impair brain function and cognition.
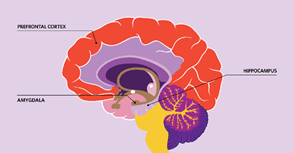
© ISTOCK.COM, JAMBOJAM
PREFRONTAL CORTEX: In some studies, people who are lonely have been found to have reduced brain volumes in the prefrontal cortex, a region important in decision making and social behavior, although other research suggests this relationship might be mediated by personality factors. Rodents that have been isolated from their conspecifics show dysregulated signaling in the prefrontal cortex.
HIPPOCAMPUS: People and other animals experiencing isolation may have smaller-than-normal hippocampi and reduced concentrations of brain-derived neurotrophic factor (BDNF), both features associated with impaired learning and memory. Some studies indicate that levels of the stress hormone cortisol, which affects and is regulated by the hippocampus, are higher in isolated animals.
AMYGDALA: About a decade ago, researchers found a correlation between the size of a person’s social network and the volume of their amygdala, two almond-shaped brain areas associated with processing emotion. More-recent evidence suggests the amygdalae are smaller in people who are lonely.
Read the full story. https://www.the-scientist.com/features/how-social-isolation-affects-the-brain-67701?_ga=2.196126316.1545883068.1610776046-339665001.1610776046

Effectiveness of the Close Collaboration with Parents intervention on parent-infant closeness in NICU
Published: 11 January 2021 He, F.B., Axelin, A., Ahlqvist-Björkroth, S. et al. Effectiveness of the Close Collaboration with Parents intervention on parent-infant closeness in NICU. BMC Pediatr 21, 28 (2021). https://doi.org/10.1186/s12887-020-02474-2
Abstract
Background
Parent-infant closeness during hospital care of newborns has many benefits for both infants and parents. We developed an educational intervention for neonatal staff, Close Collaboration with Parents, to increase parent-infant closeness during hospital care. The aim of this study was to evaluate the effectiveness of the intervention on parent-infant closeness in nine hospitals in Finland.
Methods
Parents of hospitalized infants were recruited in the hospitals during 3-month periods before and after the Close Collaboration with Parents intervention. The data were collected using daily Closeness diaries. Mothers and fathers separately filled in the time they spent in the hospital and the time of skin-to-skin contact with their infant during each hospital care day until discharge. Statistical analyses were done using a linear model with covariates.
Results
Diaries were kept before and after the intervention by a total of 170 and 129 mothers and 126 and 84 fathers, respectively. Either parent was present on average 453 min per day before the intervention and 620 min after the intervention in the neonatal unit. In the adjusted model, the increase was 99 min per day (p = 0.0007). The infants were in skin-to-skin contact on average 76 min per day before the intervention and 114 min after the intervention. In the adjusted model, skin-to-skin contact increased by 24 min per day (p = 0.0405).
Conclusion
The Close Collaboration with Parents intervention increased parents’ presence and skin-to-skin contact in nine hospitals. This study suggests that parent-infant closeness may be one mediating factor explaining benefits of parenting interventions.
Source: https://bmcpediatr.biomedcentral.com/articles/10.1186/s12887-020-02474-2
Predicting premature births with a digital health tool
PopNatal

Preterm birth, or babies born before 37 weeks, is the leading cause of newborn death. Just as concerning are babies that are born very prematurely — at 28 weeks or less — who have these risks extend into the first year of life, including being at a higher risk of SIDs. Right now, the only way to predict preterm birth is by asking a pregnant woman if she’s given birth prematurely before.
“That approach only picks up 7% of preterm births,” says Dr. Avi Patil, CEO of Nixxi. His company has developed a digital health tool, called PopNatal, to more accurately predict preterm births. “Our rate is about 75% sensitivity to pick up women who will deliver prematurely.”
PopNatal tallies up more than 300 risk factors of preterm birth for each patient. These include being pregnant with twins, triplets or more; IVF conceptions; a short time period between pregnancies; high blood pressure; diabetes; the age of the mother; smoking; drinking; stress; working long hours with long periods of standing; and family history of preeclampsia.
PopNatal consolidates those factors into an algorithm that determines whether a woman is at high risk or low risk of preterm birth. The form takes around 15 minutes to fill out, and Nixxi will send the results directly to your doctor within 72 hours.
PopNatal has been tested on thousands of pregnant women, and was developed by Patil, who is a high-risk obstetrician, and Dr. Chad Grotegut, a maternal-fetal medicine specialist. Between them, they have 27 years of experience in providing care to high-risk pregnancies.
They want it to be used broadly, not just by those with good health insurance. Currently, women who want to use it directly can access the tool online to get an individual risk assessment and companion report with guidance. Nixxi is also looking to work with health care providers.
Next up, Patil told CNET Nixxi is developing blood tests to pinpoint very high-risk women, and to pick up babies who have a higher risk of being in the NICU.
Source: https://www.cnet.com/health/at-ces-2021-baby-tech-keeps-booming/
How it Works


Genetic Markers Linked to Preterm Birth Identified
December 3, 2020
Research led by the University of Chicago has discovered two genes, HAND2 and GATA2, that could influence whether a woman gives birth prematurely or not.
“These genes are both important transcription factors that regulate the expression of several other genes,” said Ivy Aneas, Ph.D., a research associate at the University of Chicago and one of the lead researchers involved in the study.
“HAND2 mediates the effect of progesterone on the uterine epithelium while GATA2 is involved in stem cell maintenance.”
The researchers hope that these results may help clinicians predict whether preterm birth is a likely pregnancy outcome and plan accordingly.
Previous studies have suggested that there is a genetic element to preterm birth, but a lack of knowledge about gene expression in placental and endometrial cells has made it hard to pinpoint these connections more accurately.
“When you’re studying a disease, there are typically a lot of genetic and tissue resources available in public databases,” said co-senior researcher Carole Ober, Ph.D., a professor at the University of Chicago. “But pregnancy related conditions, like preterm birth, get much less attention or funding, and as a result pregnancy-relevant tissues are not well represented in those databases.”
To investigate this further, Aneas and colleagues tested endometrial cells attached to the placenta after birth for markers of gene expression. They collected transcriptome data by sequencing the RNA, searched for epigenetic modifications and evaluated chromatin structural changes. They then compared these results with genome wide association data from 56,384 women collected in a study looking at pregnancy duration.
As reported in the journal Science Advances, the team found two new genes linked with preterm birth — HAND2 and GATA2, which are involved in the process of ‘decidualization’ when the endometrial cells prepare for pregnancy and placenta formation by implanting into the uterine wall.
From the data the researchers collected, they think HAND2 is directly linked with gestational duration and in endometrial cells they think GATA2 is the target of nearby genetic variants linked with preterm birth. Neither of these genes have previously been linked with pregnancy length.
“The fact that we identified a link between these two genes and the duration of gestation suggests that their roles in pregnancy may be more important than previously anticipated,” said co-first author Noboru Sakabe, Ph.D., a researcher at the University of Chicago.
The researchers caution that they only analyzed one type of cell taken from three individuals so their results need to be replicated. The cells were also collected after birth, so they acknowledge there may be changes that occur during pregnancy that were not reflected here.
“Future studies that include fetal cells from the placenta and uterine or cervical myometrial cells could reveal additional processes that contribute to gestational duration and preterm birth, such as those related to fetal signaling and the regulation of labor,” write the authors.

Kat’s Corner

Three invaluable tools to boost your resilience | BBC Ideas
•Jul 31, 2020 Dr Lucy Hone
Resilience (A Quote, A Song and a Picture)
Kathy’s Picks:
A Quote: Do not judge me by my success, judge me by how many times I fell down and got back up again.” ― Nelson Mandela
A Song: I Hope You Dance”by Lee Ann Womack
And a picture:

All of You Preemies out there

Kat’s Picks:
A Quote: “When we learn how to become resilient, we learn how to embrace the beautifully broad spectrum of the human experience.”― Jaeda DeWalt
A Song: “Hey World (Don’t Give Up)”—Michael Franti
And A Photo:

Flysurfer Kiteboarding presents: Montenegro’s Hidden Coastline
Nov 20, 2015






